ABSTRACT
An exceptionally large, lustrous baroque-shaped South Sea cultured pearl from Indonesia was studied by high-resolution X-ray computed microtomography (X-ray µ-CT). The undrilled specimen measured 34.6 mm wide and 40.3 mm long. It was hollow and contained a loose bead. Analysis revealed a large internal cavity filled with liquid, gas, and organic material. The nacre thickness averaged 2.3 mm, and the bead measured 9.1 mm, both well within the typical ranges for South Sea cultured pearls. It is proposed that the cavity was originally occupied by spongy, water-bearing organic material, which inflated the pearl sac and provided a supporting surface for nacre deposition at a distance from the bead. After the nacre coating fully enclosed the organic material, gases were liberated and trapped inside the pearl.
INTRODUCTION
South Sea cultured pearls, cultivated using the bivalve Pinctada maxima, have an average size of about 13 mm (Strack, 2006). The shells reach sizes above 30 cm, making them the largest known pearl mussel and enabling the production of the largest commercially traded cultured pearls, with diameters occasionally above 20 mm (Strack, 2006). Currently, the primary production areas include Indonesia, Fiji, Tahiti, the Philippines, Myanmar, and the western and northern coasts of Australia. Relatively high tolerance to environmental influences (Strack, 2006) and the size of the pearls produced make this mollusk one of the most interesting species for culturing.
The cultured pearl’s colors correspond to the color of the donor bivalve’s lip (Elen, 2001; Strack, 2006), which is the epithelial tissue at the nacreous margin on the inner part of the shell. The overtone colors typically range from pink and green to blue (Gervis et al., 1992; Elen, 2001). It is common practice to reuse the cultured pearl sac several times, with successively larger cultured pearls formed. In addition, large nonbead-cultured pearls, as well as South Sea keshi pearls, occur either by chance or through cultivation (Hänni, 2006, 2007). Irregularly shaped baroque specimens are sometimes hollow or contain a loose bead. Yet they rarely reach the size of the exceptional cultured pearl described here.
X-ray computed microtomography (X-ray µ-CT) is a nondestructive method and thus ideally suited for the study of valuable pearls. Sample preparation is minimal (Wehrmeister et al., 2008; Karampelas et al., 2010), and analyses are carried out at ambient pressures. Methods requiring vacuum could cause severe damage to hollow pearls (Sturman, 2009). High-resolution digital X-ray images produced by X-ray µ-CT can be used to differentiate between aragonite and vaterite, the latter of which is responsible for the lack of luster in freshwater pearls (Wehrmeister et al., 2007, 2008). Lastly, Krzemnicki et al. (2010) showed that µ-CT radiographs are more effective than traditional radiographic images in resolving internal features of natural and cultured pearls.

MATERIALS AND METHODS
Our specimen was an undrilled, silver-colored baroque cultured pearl, with a pink to blue overtone, (figure 1) cultivated in Pinctada maxima. It was made available for analysis by the owner. The lustrous 34.6 × 40.3 mm specimen was cultured on the Indonesian island of Lombok (figure 2), which hosts a number of pearl farms in well-protected bays that provide excellent pearling conditions for Pinctada maxima (Zwaan and Zwaan, 1997).
Computed X-ray microtomography was carried out using a Procon X-Ray CT-Alpha commercial instrument at the Institute for Geosciences, Johannes Gutenberg University. The device and methods were largely identical to those used by Karampelas et al. (2010), though optimized using an accelerating voltage of 100 kV and a target current of 120 µA. The beam was pre-filtered by a 1.0 mm thick aluminum foil. The sample was mounted on a rotation stage at a distance of 250 mm from the X-ray tube for optimal magnification and resolution. We recorded 800 projections over 360°, with an average of 10 images per projection and an acquisition time of 1.5 seconds per image. This resulted in a dataset with 22.4 µm per voxel (volume pixel) spatial resolution. Data processing was carried out with Octopus 8.5 commercial software and Avizo Fire 7.0 3D analyzing software.
RESULTS
Figure 3A shows a large cavity that occupies most of the specimen’s interior. The bead is 9.08 ± 0.02 mm in diameter and loose (i.e., not in direct contact with the underlying nacre). Based on 18 measurements, the nacre thickness varies between 0.9 and 3.3 mm, with an average thickness of 2.3 ± 0.7 mm. Along much of the interior, narrow layers of nacre protrude inward into the large cavity (figure 3A, yellow arrow). Two irregular nacre regions were observed protruding outward; one has a very thin nacre coating of 0.9 mm. The thicker one is shown in figure 3A, and indicated with a white arrow.
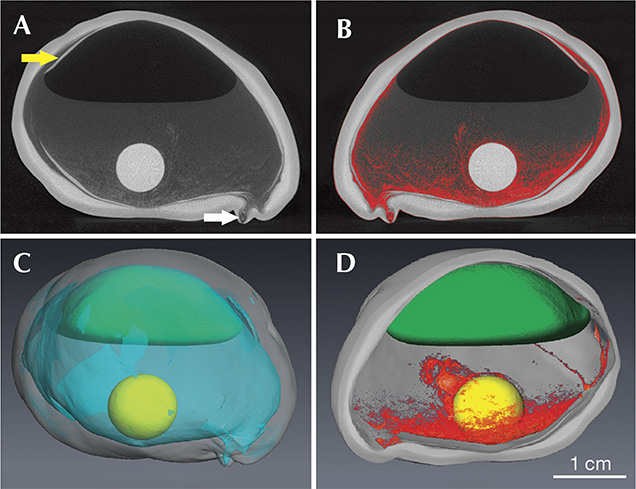
Apart from the nacre and bead, which have high X-ray densities and appear light gray in figures 3A and 3B, several different phases with distinct X-ray densities could be distinguished by their grayscale values within the cavity. Phase 1, with the lowest X-ray density (dark gray or black appearance), was situated in the uppermost part of the cultured pearl. This phase was separated from phase 2 by a meniscus in the lower portion. Phase 3 was spread out across the lower part and the sides of the cavity (red in figure 3B), providing a base and partial cover for the bead. Judging from its grayscale similarities to the ambient air around the specimen, phase 1 is interpreted to be a gas phase. Phase 2, on the other hand, showed a meniscus toward phase 1, which is typical for a liquid-gas interface. The noise produced from shaking the pearl confirmed that this phase was a liquid. Phase 3 had grayscale values between that of phase 2 (liquid) and the nacre. It had a spongy appearance (red in figure 3B) and extended in finger-like shapes above and partially along the bead. This phase likely represented organic material. No inorganic phase was observed in this area. In total, phase 2 (liquid), with a volume of approximately 6 mL (table 1), was the most abundant phase after the nacre, while phase 1 (gas) occupied approximately half the space of the liquid phase. Phase 3 (organic) was the least voluminous phase.
| Table 1. Relative abundance of the different phases of the cultured pearl specimen. a | ||
|---|---|---|
| a Volumes assigned to specific X-ray grayscales of the various parts of the specimen were calculated by software. As the cultured pearl contains multiple dimples, the volume was found to be less than that of an ideal ellipsoid. | ||
| Volume (cm3) | Vol. % | |
| Nacre | 7.72 | 44 |
| Phase 1 (gas) | 3.40 | 19 |
| Phase 2 (liquid) | 6.06 | 34 |
| Phase 3 (organics) Total cavity (1+2+3) | 0.19 9.65 | 1 54 |
| Bead Total pearl | 0.39 17.76 | 2 100 |
With the current state of analytical instruments, no further nondestructive methods could be employed to identify the content of this sample. One destructive option would be to release the liquid and gaseous contents for analysis by drilling (with gas chromatography, for instance). This would risk severely damaging the sample, however.
DISCUSSION
This specimen was far above average size, as most Australian and Indonesian baroque cultured pearls range from 12.5 to 13.0 mm (Strack, 2006). Only a small percentage of baroque-shaped South Sea cultured pearls are known to have reached 40 mm in size (Strack, 2006), but none have matched the total volume of the example studied here. The specimen’s average nacre thickness of 2.3 mm fell within the average South Sea range of approximately 0.5–4.0 mm (Strack, 2006). This indicates nacre growth rates comparable to those of other South Sea cultured pearls and shows that the specimen’s large size was not caused by abnormally fast growth. The bead also compared favorably with the range of bead sizes commonly used for the first grafting of Pinctada maxima (Strack, 2006). Therefore, the characteristic differences between this specimen and other baroque cultured pearls were its unusually large size and the exceptional volume of its cavity. Table 1 shows that the cavity measured 9.65 cm3, comprising slightly over half of the total volume.
Templates for Nacre Deposition. A beaded pearl is grown by inserting a bead into the mussel tissue or gonad, along with a tissue graft that has the potential to secrete nacre. These tissue cells subsequently multiply into a pearl sac that eventually encloses the bead and generates a sheltered compartment for the creation of the pearl (Akamatsu et al., 2001; Landmann et al., 2001). In most cases, a cultured pearl’s shape and size depend heavily on the shape and size of its bead. From our observations and from those of others, nacre coating of the bead usually starts with a thin layer of organic material deposited on the bead, followed by regular nacre secretion until the pearl is harvested (Caseiro, 1993; Jacob et al., 2008). Round shapes are obtained when the bead, the organic layer, and the nacre are in contact with each other, providing a more stable surface (a so-called template) for the regular deposition of nacre layers. In cases where the bead is removed—upon rejection by the bivalve, for instance—the pearl sac collapses and flat pearls are formed. This specimen showed the opposite effect from a very rare instance of an inflated pearl sac; in this case, an exceptionally large cultured pearl was formed without bead removal when the pearl sac inflated.
The pearl contained a loose bead, much smaller than the pearl itself, and thus the nacre coating could not have been formed in contact with the bead. Similarly hollow pearls filled with liquid and smaller organics are known, occurring both naturally and from culturing (Sturman, 2009). The latter sometimes display loose beads as well.
Although a solid surface is generally required for nacre deposition, nonbead-cultured pearls demonstrate that the surface does not have to be rigid. The center of a nonbead-cultured pearl typically contains an organic “core,” which can be used to distinguish them from natural pearls (Hänni, 2006; Karampelas, 2010). This organic core represents a striking analogy to the first layer of organic material deposited on the bead in beaded cultured pearls. We propose that this organic material was not an accidental inclusion in addition to the bead; rather, it represented the organic template. It is regularly produced by the pearl sac at the start of nacre deposition in beaded pearls as well (Jacob et al., 2008), and we propose that this organic material expanded and supported the formation of a pearl sac much larger than normal. This process may bear some similarities with a new development seen in cultured pearl production. Cartier and Krzemnicki (2013) describe the use of a newly developed organic nucleus that is highly absorbent. Initially compact, it grows into a large gelatinous nucleus after insertion into the gonad of the mussel. Since the tissue graft stays in contact with the swelling organic nucleus, this process results in an inflated pearl sac, eventually producing large baroque pearls (the first generation containing the gelatinous nucleus is not sold, according to the authors). A critical difference between this novel culturing method and the hypothesis proposed for our specimen is that we infer an expanded organic layer coating of the bead nucleus rather than an expanding nucleus.
Thus, we propose that the nacre in this exceptionally large cultured pearl was deposited on an organic template that forms regularly as the first product of nacre secretion. To support such a large pearl, the organic template must have originally occupied much of the space between the bead and the nacre, as seen in figure 3. This clearly represents a departure from normal conditions.
Figure 3A shows some layers of nacre flaking off toward the interior (see the yellow arrow). We believe it is an effect of nacre deposition on an unstable template provided by organic material with little support from the bead.
Hollow Pearls with Organic Fillings. Our evidence suggests that the pearl’s interior was filled with a spongy mixture of organic material (perhaps tissue) and water that provided physical support for the deposition of the nacre coating. This also indicates that the pearl sac did not fully enclose the bead, meaning it did not heal completely after surgery, providing an opening for water to enter. Net-like remnants of the organic phase could still be seen enclosing the bead (figure 3D). It is likely that some of this material was introduced upon grafting and due to proliferation following a bacterial infection or other accidental inclusions or contamination of tissue material.
In due course, the organic material within the nacreous coating decayed, liberating foul-smelling gases that collected at the top of the pearl (figure 3C). Indeed, “organic odors” have been detected upon drilling or otherwise breaking open hollow pearls (Sturman, 2009).
The pearl had a small area where the nacre coating was only 0.9 mm thick (figure 3). This area probably represents a perforation in the nacre coat, which could have allowed the liquids and gases to penetrate or leak from the pearl. The average growth rate for nacre in South Sea pearls is approximately 1 mm per year (Strack, 2006). Assuming normal growth rates for this very thin area, the perforation must have remained open throughout most of the growth period of this remarkable specimen.
CONCLUSION
Nondestructive analysis of this exceptional cultured pearl by X-ray µ-CT revealed a multi-phase cavity filled with liquid, gas, and organic material. The cavity measured approximately four times larger than the bead in diameter, which raises the question of how the nacre coating was physically supported during pearl growth. The model presented here argues that the organic material played an important role. We submit that pathological proliferation of tissue material after grafting, possibly due to a bacterial infection, inflated the pearl sac while the enclosed spongy organic material acted as a template providing support for pearl growth. Once enclosed in nacre, the organic material decayed, liberating gases that had collected inside the pearl.
The proposed model may well represent a viable process for the formation of hollow pearls. If bacterial infections are involved, such products represent true “survivors,” underscoring the exceptional nature of this large South Sea cultured pearl.
Ms. Otter is a graduate student at the Institute of Geosciences, Johannes Gutenberg University, in Mainz, Germany. Drs. Enzmann and Wolf are researchers in charge of the laboratory for micro-computed tomography at the Institute of Geosciences. Dr. Wehrmeister is a research scientist at the Institute of Geosciences. Dr. Jacob (dorrit.jacob@mq.edu.au) is associate professor and ARC Future Fellow at the Department of Earth and Planetary Sciences, Macquarie University in North Ryde, Australia.
Ms. Otter is a graduate student at the Institute of Geosciences, Johannes Gutenberg University, in Mainz, Germany. Drs. Enzmann and Wolf are researchers in charge of the laboratory for micro-computed tomography at the Institute of Geosciences. Dr. Wehrmeister is a research scientist at the Institute of Geosciences. Dr. Jacob (dorrit.jacob@mq.edu.au) is associate professor and ARC Future Fellow at the Department of Earth and Planetary Sciences, Macquarie University in North Ryde, Australia.




