ABSTRACT
To improve the accuracy and efficiency of trace element analysis in ruby and sapphire, standards sets containing matrix-matched corundum with targeted levels of beryllium, magnesium, silicon, titanium, vanadium, chromium, iron, and gallium were created. Ultra-high-purity corundum was also incorporated as a “true matrix zero.” To our knowledge, these sets contain the most accurate standards for the key trace elements in corundum today. The standards were designed to cover the optimal range of the main trace elements typically characterized in natural ruby and sapphire. Additionally, we were able to accurately quantify silicon in corundum by successfully resolving the 28Si+ peak from the 27Al1H+ peak using the superior mass resolving power of secondary ion mass spectrometry (SIMS) as compared to laser ablation–inductively coupled plasma–quadrupole mass spectrometry (LA-ICP-QMS).
INTRODUCTION
To correctly assign geographic origin (Emori and Kitawaki, 2015; Sutherland et al., 2015), formation history (Peucat et al., 2007), and color mechanisms (Emmett et al., 2003) in ruby and all colors of sapphire, accurate key trace element data is an essential component. As magnesium, titanium, vanadium, chromium, iron, and gallium are found in all natural sapphire in varying amounts, these trace elements are identified as the key trace elements for natural corundum. Additionally, beryllium is significant to quantify, as the presence of this trace element can indicate treatment. The various quantitative trace element detection techniques used today for analyzing mineral species—electron microprobe, X-ray fluorescence (XRF), secondary ion mass spectrometry (SIMS), laser-induced breakdown spectroscopy (LIBS), and laser ablation–inductively coupled plasma–mass spectrometry (LA-ICP-MS)—require standards for each trace element of interest. Essentially, these sophisticated and expensive instruments that allow us to quantify the trace elements in gems can only be as accurate as the standards used. With gemological laboratories issuing origin reports for ruby, sapphire, and other gems, it is essential to have a high level of certainty associated with the quantitative determination of trace element chemistry. The standard’s “matrix,” the material that it is comprised of, matters a great deal: Matching both the composition and structure of the standard to those of the sample being analyzed will yield the most accurate results (Sylvester, 2008). For example, trace levels of vanadium in ruby (or any of the colors of sapphire) are most accurately quantified by comparing against a piece of single-crystal corundum with a known level of vanadium in it as opposed to some other material containing vanadium, such as silicate glass.
However, commonly used standards such as those produced by the National Institute of Standards and Technology (NIST) are generally not available in matrix-specific forms, so silicate glasses such as NIST Standard Reference Materials (SRM) 610 and 612 are frequently applied. As the silicate glass matrix differs significantly from the crystalline alumina matrix of sapphires and rubies in both composition and structure, what is known as a “matrix effect” can occur. This effect leads to inaccuracy for many of the key trace elements characterized. A potential outcome of calibrating an instrument with a matrix different from the sample to be measured is the enhancement or suppression of trace element levels detected (Strasheim and Brandt, 1967). Furthermore, NIST SRM 610 and 612 glasses are not certified for many of the trace elements they are being used for, as is the case for corundum, and both have been shown to demonstrate variability (Pearce et al., 1997). Of the key trace elements normally analyzed in corundum, these glasses are only certified for iron and chromium (certificates of analysis from NIST are available at https://nemo.nist.gov/srmors/certificates/view_certGIF.cfm?certificate=610 and https://nemo.nist.gov/srmors/certificates/view_certGIF.cfm?certificate=612). Another drawback to the NIST glasses for corundum: Aluminum is present at low concentrations (around 1 wt.%) in the NIST glasses but is a major element in corundum (around 52.9 wt.%). The projection from low concentration to high concentration from the standards to unknowns can introduce significant error—projections work best when similar concentrations are compared between standard and unknown.
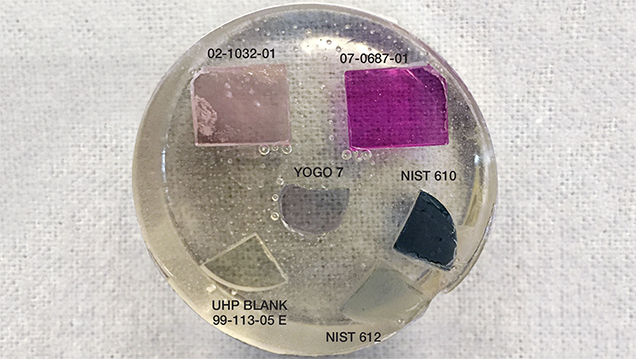
Due to concerns regarding composition and structure matrix effects, GIA decided years ago to generate corundum standards for its LA-ICP-MS instruments (Wang et al., 2006). After a decade of use for both production and research applications, new standards sets have been created to further improve accuracy (discussed in the Background section) and efficiency. To optimize the new standards sets for regular laboratory use, each individual set contains a minimum number of standards (in our case, four pieces of corundum and NIST SRM 610 and 612 glasses embedded in epoxy—see figure 1), reasonably capturing the range of interest for each key trace element to be quantified. As there is a need to characterize many samples routinely for origin reporting, efficiency is paramount. We reduced calibration time by combining as many trace elements as possible into single standards. We accomplished this by growing “multi-doped” synthetic corundum crystals. Instead of having only one or two of the trace elements of interest in a single standard, we worked with FLIR's crystal growth group, Scientific Materials of Bozeman, Montana, to produce highly uniform Czochralski-grown single crystals of corundum containing up to six intentionally doped trace elements (figure 2). To incorporate beryllium into the standards, slices from one of these custom-grown crystals were diffused with beryllium for 100 hours at 1800°C in pure oxygen, generating one standard in which seven trace elements can be calibrated at the same time. Beyond reducing instrument calibration time, this approach of fewer individual measurements in the calibration cycle also enhances accuracy, precision, and repeatability, as there is less chance for instrument signal drift, and therefore more accurate conversion of instrument signal into trace element concentrations.

Even with this approach, it was impossible to incorporate all of the trace elements of interest into the synthetically grown crystals at levels that best represented what is found in natural corundum. As we were unable to produce a standard for mid-range to higher iron levels and moderate silicon concentrations with Czochralski growth, we selected a series of natural sapphires from Yogo Gulch, Montana.
BACKGROUND
Original and Next-Generation Corundum Standards Sets. The first corundum standards sets GIA created for its LA-ICP-MS instruments focused on accuracy by using matrix-matched standards. These sets consisted of seven different corundum standards: one high-iron natural sapphire and six synthetically grown sapphire crystals with either one or two intentionally doped trace elements each. A need for increased accuracy, better targeting of trace element levels in natural ruby and sapphire, and enhanced efficiency led to the development of the current generation of standards sets. To this end, we focused on the following criteria while minimizing uncertainty:
- Using corundum that clearly displays highly uniform levels of the trace elements of interest
- Capturing the appropriate ranges of the trace elements in our standards by matching them to levels occurring naturally in sapphire and ruby
- Developing a high-purity blank sample to represent the absence of trace elements in corundum
- Using the most accurate methods available for corundum as the basis of our calibration work
Uniformity of Trace Elements in the Standards. For a standard to produce accurate and reproducible results, it must be extremely homogenous (even more so for a spot method such as LA-ICP-MS than for a bulk method). Czochralski-grown synthetic crystals of corundum and other matrices are known for their exceptional crystal quality, including highly uniform distributions of trace elements. Demand in the 1960s for ruby crystals suitable for lasers led to the refinement of the Czochralski technique for the production of commercial solid-state lasers (Harris, 2004). For this reason, we chose the Czochralski technique as our growth method. As mentioned above, producing Czochralski-grown corundum with mid-range to high levels of iron and moderate levels of silicon was not practical, so we sourced extremely uniform Yogo sapphire crystals (shown in figure 3) from Eric Braunwart of Columbia Gem House (Vancouver, Washington). Surprisingly, we found with SIMS that in addition to iron, several of the other key trace elements of interest for corundum—including Mg, Si, Ti, and Ga—were as uniformly distributed in the natural Yogo sapphires as they were in the Czochralski-grown material. We have confirmed that silicon and nickel are also present in Yogo sapphires in amounts up to 26 and 12 ppma, respectively, and are likewise homogenously distributed.

Targeting the Trace Element Levels in the Standards. To best determine the concentrations of trace elements in unknown samples, it is optimal to have standards that contain similar to somewhat higher levels of the same trace elements. We attempted to create standards with concentrations at the 50th percentile or higher of the key trace elements found in natural sapphire and ruby based on data from the GIA research collection. GIA has been gathering and cataloging natural ruby and sapphire from identified sources worldwide as part of its reference collection effort. The purpose of this collection is to carefully document material from each accessible source and to produce detailed information on the inclusions and trace element chemistry by location (Chapin et al., 2015; Pardieu and Vertriest, 2016). This collection contains over a thousand samples and is one that GIA adds to each time its field gemologists visit a unique mining area of commercial interest or significance. The collection includes basalt-related sapphire, metamorphic sapphire, marble-hosted ruby, high-iron ruby, and additional types of corundum beyond these four categories. The samples measured include gem and non-gem quality, and for the key trace elements, the data was quantified using the original corundum standards sets. For our purposes, we are limiting the discussion to these key trace elements that are found in the corundum matrix, and not those found in inclusions.
Using data from the GIA reference collection, we considered the entire range of each key trace element detected, with the 50th and 75th percentiles presented in table 1. It is worth noting that the highest levels detected can be considerably higher than the 75th percentile levels reported. For beryllium, where we were interested in detecting diffusion treatment (Emmett et al., 2003), we targeted around 60 ppma, a level sufficient to distinguish between diffused samples (where the surface of the gem can show significant beryllium levels) and undiffused samples (with beryllium levels that are undetectable or very low, such as 1–2 ppma).
Introduction of a Matrix-Matched Zero. The new standards sets address accuracy in part by incorporating a true matrix-matched “zero”—a piece of ultra-high-purity sapphire containing essentially no detectable trace elements. Using this approach of defining the zero signal point for each of the trace elements as the pure matrix instead of a background of solely helium gas minimizes matrix effects, as interferences will be eliminated and the zero point of the calibration line will now be far more accurate (Lin et al., 2016). Standard undoped colorless sapphire and high-purity alumina powders used commercially still contain several ppmw of various trace elements such as chromium, titanium, magnesium, and iron. For example, high-purity alumina with a 5N designation (meaning “five nines,” or 99.999% purity) still contains up to 10 ppmw combined trace elemental impurities. For our purposes, it was important to obtain undoped sapphire of significantly higher purity—ideally 6N or better. To accomplish this, the most refined single-crystal sapphire known to us was acquired from Chandra Khattak of the former Crystal Systems Corporation (sold to GT Advanced Technologies in 2010). The single-crystal sapphire was material that had been recrystallized multiple times using the heat exchanger method (HEM) to further reduce the impurity levels found in the original aluminum oxide raw material (Khattak and Schmid, 2001).
Ensuring Accuracy. As mentioned earlier, the accuracy of the measurements obtained from standards-dependent quantitative analytical techniques employed by many research laboratories is limited by the adequacy of the standards used. To improve the accuracy of the new standards sets over the original sets, many steps were taken to develop a highly accurate calibration methodology with minimal combined uncertainty (figure 4). We sought a method based on first principles for determining the levels of the trace elements of interest in our standards. In other words, we wanted a way of determining trace element levels in corundum that did not require referencing the levels of those same trace elements in some other standard (i.e., a noncircular method). This led us to an approach used by the geology community in which individual ion implant doses verified by Rutherford backscattering spectrometry (RBS) are used to calibrate the SIMS apparatus prior to measuring final samples (Burnett et al., 2015). As the concentrations of the ion implants arrived at from the voltage and current settings are accurate to only about 30% (Burnett et al., 2015)—an unacceptable level of uncertainty for our purposes—the actual dosing levels achieved for each ion-implanted wafer were determined through multiple RBS characterizations (or for the lighter isotopes 9Be, 24Mg, and 28Si where this was not practical, through a method described below). RBS is a nondestructive first-principles (standardless) quantitative analytical technique that is capable of 1% absolute accuracy (Colaux and Jeynes, 2014). SIMS was chosen as our technique for performing the ultimate calibration work on the final corundum standards, as it is a precise technique with fine mass-resolving power capable of quantifying all of the elements of interest to us.
As trace elements are not distributed with absolute uniformity in synthetic sapphire, and each natural Yogo sapphire slice will have its own composition, nine points on all individual pieces for each standards set were characterized by SIMS (see figure 4). Simply put, the same standard type in each standards set has its own unique set of values. This is quite a departure from the SRM NIST 610 and 612 glasses, where most researchers refer to published tables for the concentrations of the trace elements (certified and uncertified) contained within any piece of a particular series of NIST glass, of which there are thousands of pieces worldwide (Pearce et al., 1997). For example, the chromium level in a Czochralski-grown corundum crystal is nearly uniform from top to bottom, but its distribution coefficient will result in pieces from the bottom of the crystal having levels slightly lower than those from the top. To label the entire crystal as having a single chromium concentration would result in inaccuracy. As SIMS can easily quantify not only differences between the top and bottom of the crystal, but also point-to-point differences across areas less than one square millimeter, we made sure to quantify the levels of chromium (and all of the other trace elements) individually for each separate standard piece from each source. These characterized standards were now ready to be assembled into finished sets for GIA’s LA-ICP-MS instruments.
To most accurately report data based on our new corundum standards, it is necessary to calculate the total uncertainty in the final values for each trace element in each standard, factoring in all measurement and instrument errors and uncertainties. We accounted for all of the sources of errors and uncertainties to the best of our ability and combined them via the root sum of the squares method following the NIST-referenced INC-1 (1980) recommendation reached by the Working Group on the Statement of Uncertainties, approved by both the International Committee for Weights and Measures (CIPM) and the International Bureau of Weights and Measures (BIPM).
MATERIALS AND METHODS
Crystals for Standards. Crystal Growth. As mentioned, we selected the Czochralski method, known for producing crystals of high quality with uniform doping of trace elements, to grow synthetic sapphire crystals containing the key trace elements for our standards. After many crystal growth runs to best understand the doping characteristics of each trace element of interest, two final crystals were grown by Scientific Materials to provide material to be used in our standards sets. These two samples were both doped with the trace elements Mg, Ti, V, Cr, Fe, and Ga. Crystal 02-1032 had lower concentrations of Mg, Ti, and Cr and higher concentrations of V, Fe, and Ga; crystal 07-0687 had lower concentrations of V, Fe, and Ga and higher concentrations of Mg, Ti, and Cr (figure 2 top and bottom, respectively). Pieces from crystal 02-1032 were subsequently diffused with Be for a period of 100 hours at 1800°C in pure oxygen to provide a standard for this trace element. From each of the two boules, seven polished slices measuring 8 × 10 × 2 mm were fabricated by Scientific Materials. These seven pieces from each boule constitute only a small fraction of the total available material for the two boules.
Both crystals were grown directly from the melt at approximately 2050°C and pulled from iridium crucibles using seeds at an orientation 60° off the c-axis (the growth direction was perpendicular to the n-plane, to facilitate growth of more defect-free material). The raw materials used were 5N purity powders of Al2O3 and dopant metal oxides. After the powders were loaded into iridium crucibles, the crystals were grown in a nitrogen atmosphere containing 3,000 ppmw oxygen. The crystals were pulled at an extraction rate of 0.015 inches (0.38 mm) per hour. The final size of crystal 02-1032 was 40 mm in diameter by 150 mm in length at diameter. The final size of crystal 07-0687 was 30 mm in diameter by 150 mm in length at diameter.
Magnesium, titanium, and vanadium were “well-behaved ions” under the growth conditions used, in that they did not exhibit vaporization and their incorporation into the crystals was predictable based on our knowledge of their distribution coefficients. For chromium, iron, and gallium, some vaporization was noted and they were less predictably incorporated into the grown crystals under the conditions used. Therefore, maintaining well-controlled growth conditions to reduce variability was critical.
Ultra-High-Purity Corundum. The ultra-high-purity corundum was obtained from the former Crystal Systems, which employed the HEM technique for producing UV-grade sapphire. To enhance the purity of the material, the sapphire was recrystallized multiple times (C. Khattak, pers. comm., 2003). Ultimately, we determined a combined total ppma level of less than 0.5 for the key trace elements in this material. Seven finished pieces were fabricated from this material by Scientific Materials.
Yogo Sapphire. Several polished slices of highly uniform light blue Yogo sapphire crystals were carefully selected by Eric Braunwart of Columbia Gem House. We obtained seven for our standards sets. Yogo sapphires are known for mid-range to high iron content and exceptional color uniformity (Mychaluk, 1995; Emmett et al., 2017), which we expected would correspond to trace element uniformity (figure 3).
Standards Characterization. Our pursuit of accuracy led us to create the standards sets using a two-step approach: First, create highly controlled and measurable standards (ion implants) to calibrate extremely precise instrumentation (SIMS), and then use the standardized SIMS to calibrate selected corundum pieces to be incorporated into final product standards sets (see figure 4). The steps taken to create the SIMS calibration standards and then the final product corundum calibration standards for GIA’s LA-ICP-MS instruments are listed below.
Ion Implant Standards Creation. The first step was to create a separate calibration standard for each trace element of interest in a corundum matrix. This was accomplished at INNOViON Corporation (San Jose, California) using ion implantation, a method that allows one to precisely control the concentration (dose) and depth profile of a specific amount of a trace element to be placed into a matrix (Ryssel and Ruge, 1986). Single isotopes of 9Be, 24Mg, 28Si, 48Ti, 51V, 52Cr, 56Fe, and 69Ga were implanted separately into their own two-inch-diameter high-purity corundum wafer on top of a four-inch-diameter silicon wafer (except in the cases of 24Mg and 28Si, in which their two-inch-diameter corundum wafers were placed on top of four-inch-diameter gallium arsenide wafers). The four-inch silicon or gallium arsenide wafers functioned as witness samples for the ion implantation process. The target dose (or “fluence”) for each isotope was 1.00 × 1015 at/cm2 (except 24Mg, which targeted a 1.00 × 1014 at/cm2 dose). The implant energy used for our wafers was 200 KeV for all isotopes except 9Be, which used an energy of 100 KeV. For all wafers, the tilt angle between the ion beam and the wafer surface normal was 7°.
Verification of the Ion Implant Dosing. For the key trace elements that are significantly heavier than the matrix elements aluminum and oxygen (Ti, V, Cr, Fe, and Ga), RBS was used to determine the actual dosing. The RBS technique can determine the concentrations with depth of isotopes in a matrix of lighter elements by bombarding a sample with a beam of high-energy particles and measuring the number and energy of backscattered ions (Verma, 2007). For those key trace elements that are close in mass or lighter than aluminum and oxygen (Be, Mg, Si), the ion implant dosing had to be verified by a consensus-dosing approach using comparisons to well-characterized existing implants.
The implant doses of the heavier isotopes 48Ti, 51V, 52Cr, 56Fe, and 69Ga were characterized by RBS at Evans Analytical Group (EAG) in Sunnyvale, California; the Ion Beam Laboratory at the University at Albany, State University of New York (SUNY); and the Tandetron Accelerator Laboratory of the University of Western Ontario (UWO) in London, Ontario. For all measurements, a He++ ion beam with energy between 2 and 3 MeV was used.
To characterize the ion implant standards lighter than or close in mass to Al (9Be, 24Mg, and 28Si), EAG determined “consensus doses” (doses based on several verifications) using SIMS and their multiple reference standards for Be in Si, and Mg and Si in GaAs. When these lighter ions were implanted in their sapphire substrates, they were simultaneously implanted into larger-diameter “witness” substrates of silicon or gallium arsenide that the sapphire substrates were placed on top of. These witness samples were then measured with SIMS and directly compared to the existing reference standards produced and used by EAG. These standards served as calibrators for the SIMS to determine the doses in our witness samples. For Be, Mg, and Si, EAG had three, four, and five existing standards, respectively. We assumed that the doses in the witness samples were identical to those in the actual sapphire substrates, which was shown to be true for the heavier implants for titanium, vanadium, chromium, iron, and gallium that each had silicon witness samples under them during the implantation process.
Calibration of the SIMS and Characterization of the Final Standards. SIMS is a highly sensitive and precise quantification technique for elements from Be to U (Benninghoven et al., 1987). The technique produces elemental depth profiles by sputtering samples with a primary beam of either oxygen or cesium ions and then analyzing the secondary ions formed with a mass spectrometer. Once the doses in the ion implant standards were accurately characterized, these standards were measured with SIMS under a controlled set of conditions to determine “relative sensitivity factors,” or RSF values, for each trace element of interest to be characterized in corundum (Laufer et al., 2011). The RSF simply converts the SIMS signal for a particular trace element in a defined matrix under defined conditions into concentrations (see box A for a more detailed description). Our determined RSF values were then applied to data collected on the pieces of corundum that are now in our corundum standards sets under the same controlled set of operating conditions to accurately quantify these same trace elements. The RSF values for each trace element were determined from a series of two to four spots on the appropriate ion implant standard, verified in two separate measurement sessions on nonconsecutive days. The concentration values with combined uncertainties for each piece of corundum incorporated into the standards sets were obtained with SIMS based on averaging nine spots per piece.
| Box A: SIMS RSF and Trace Element Concentration Determination |
| With the RBS “dose” or “fluence” values Ψ (units of atoms/cm2), SIMS RSF values—the conversion factors between SIMS data for a particular element in a sample and its concentration—can be determined in the following way. We first sputter the ion implant standards with SIMS to collect the counts of the implanted ion and matrix ion. This implanted ion, or “ion of interest,” is also referred to as the “secondary ion of interest.” We express the ion counting rate of the implanted isotope as I(x)1, which is also the current of the implanted isotope. Similarly, the ion counting rate of the matrix isotope—in the case of a corundum matrix this would be 27Al+—is expressed as I(m)1, which is also the current of the matrix isotope. The RSF (units of atoms/cm3) can be determined using the relationship |
| RSF = Ψ/∫(I(x)1/ I(m)1)dx |
| where dx is the depth profile in angstroms and is determined by the product of the sputtering rate (S) in angstroms per second and the time interval in seconds recorded during the SIMS analysis: |
| dx = Sdt |
| The sputtering rate is the ratio of the simulated peak depth to the peak time in seconds: |
| S = Peak depth/Peak time |
| We attempted peak depth determination by both physical measurements and calculation with “Stopping and Ranges of Ions in Matter” (SRIM). Because of the small non-flat-bottom craters in sapphire, large uncertainties exist in physical depth measurements. The peak depths are currently calculated with SRIM, before more accurate depth measurements can be achieved. Once the RSF values for each trace element in the matrix have been determined, the atomic concentrations—n(x)—of these trace elements in other samples can be determined using the product of the RSF with the ratio of the secondary ion counting rates: |
| n(x) = RSF × (I(x)2)/(I(m)2) |
| where I(x)2 is the secondary ion counting rate, or current of the trace element isotope in a sample, and I(m)2 is the secondary ion counting rate, or current of the matrix isotope—in our case, 27Al+. |
SIMS Instrument Description and Operating Conditions. The CAMECA IMS 7f-GEO at Caltech (Pasadena, California) is a magnetic sector type SIMS instrument with dedicated options for geoscience applications. Modified from the CAMECA IMS 7f-Auto, it is equipped with double secondary Faraday cups for high-precision stable isotope analysis. The primary column consists of a duoplasmatron source; a microbeam Cs ionization source; a primary beam mass filter; fully automated slits and apertures; ion optics (lenses, stigmators, and double deflectors) for primary beam focusing and rastering; and a high-accuracy primary Faraday cup combined with a fast beam blanking system for monitoring the primary beam intensity during measurements. The secondary ion optics are continuously adjustable from –10 to + 10 kV, with a continuously adjustable imaged field and a dynamic transfer system that optimizes transmission for an analyzed area up to 500 × 500 µm2. The Nier-Johnson double-focusing mass spectrometer has fully adjustable position and width on automated entrance and exit slits, automated contrast and field apertures, a spherical electrostatic analyzer for energy filtering, an automated continuously adjustable energy slit, and a laminated electromagnet (120 mm radius) for fast peak switching. The detection system consists of an electron multiplier, double Faraday cups, and a micro-channel plate assembly.
RSF values for the elements of interest in corundum were determined through SIMS depth profiling of the implant standards. Because sapphire is an insulator and depth resolution is not a concern for this task, an O– primary beam, which can minimize sample charging during measurement, was used to acquire depth profiles of the implant standards. Matched analytical conditions were then used to determine the element concentrations of the non-implant samples. Specifically, the O– primary beam current has a potential of –13 KeV, a current of approximately 15 nA, and a size of about 15 µm. Positive secondary ions of 8.5 KeV were generated by the primary beam, rastering an area of 50 × 50 µm2 on sample surfaces. To avoid crater edge effects, a field aperture was inserted on the secondary ion path so that only the signals from the center 20 µm2 area were detected. The positive secondary ions collected were 9Be, 24Mg, 27Al, 28Si, 46Ti (samples), 48Ti (implants), 51V, 52Cr, 56Fe, 69Ga (implants), and 71Ga (samples). All secondary ions except 27Al were measured with an electron multiplier (EM). 27Al was measured with a Faraday cup because of its high intensity. The reason for measuring 46Ti instead of 48Ti on samples is to avoid possible unresolvable interference from 48Ca (0.187% natural abundance of all Ca), whereas the isotopic abundance of 46Ca is only 0.004%. The reason for measuring 71Ga instead of 69Ga on samples is to avoid possible unresolvable interferences from 56Fe13C, 57Fe12C, and 53Cr16O, as Fe and Cr are both potentially present in the samples. Because of the high 48Ti fluence of the implant standard, the possible 48Ca interference has no significant contribution to its 48Ti depth profiling results. The mass spectrometer of the SIMS was set at a mass resolving power (MRP) of 3,000 for all the measurements, which is sufficient to resolve all possible interferences to the masses of interest, especially the 27Al1H+ interference (mass = 27.98936 unified atomic mass units, or amu) to 28Si+ (mass = 27.97692 amu) with an MRP of 2,251, which is significant due to the abundant Al in sapphire (as shown in figure 5). The lowest concentrations in ppma we could detect with confidence for the key trace elements were 0.0003 for Be, 0.0002 for Mg, 0.2 for Si, 0.007 for Ti, 0.007 for V, 0.003 for Cr, 0.0006 for Fe, and 0.0005 for Ga.
Testing Standards and Samples with LA-ICP-MS. To examine the differences in using NIST glasses versus our new corundum standards for LA-ICP-MS calibration, we used a Thermo Fisher Scientific iCAP Qc ICP-MS and a New Wave Research UP-213 laser ablation unit with a frequency-quintupled Nd:YAG laser running at 4 ns pulse width. Ablation was achieved using a 55-µm-diameter circular laser spot, a fluence (energy density) of approximately 10 J/cm2, and a 15 Hz repetition rate. The internal standard used was 27Al with a value of 529,200 ppmw calculated and rounded from pure corundum. The dwell time of each laser spot was 40 seconds. The lowest concentrations in ppma we can detect with confidence by LA-ICP-QMS are 0.5 for Be, 0.1 for Mg, 0.5 for Ti, 0.05 for V, 0.2 for Cr, 3.5 for Fe, and 0.01 for Ga.
RESULTS AND DISCUSSION
Verification of the SIMS Calibration Standards. A summary of the consensus SIMS results from EAG for the actual doses of 9Be, 24Mg, and 28Si as well as the RBS results from the three labs (EAG, SUNY, and UWO) for the actual doses of 48Ti, 51V, 52Cr, 56Fe, and 69Ga in corundum are given in table 2. All values are presented with their combined uncertainties in terms of actual values and as a percentage of the determined doses. It is clear that verifying the ion-implanted doses was important in that the actual doses varied from the target doses from as little as zero to as much as 32%. The combined standard uncertainties at this point contain the uncertainties in the measured fluence values with each RBS measurement (or consensus SIMS measurement) and in calculating the mean values from the multiple RBS measurements for each fluence.
Determined RSF Values for SIMS. The RSF values with their relative standard deviations (RSD) are given in table 3. These values allow us to accurately calculate the actual concentrations of the key trace elements in our final standards originating from the custom-grown multi-doped crystals, the Yogo sapphire slices, and the ultra-high-purity blank sapphire.
Measured Values for the Final Corundum Calibration Standards. Using our RSF values, we calibrated the SIMS apparatus and determined the concentrations of the key trace elements in our new corundum standards based on nine separate points each. The ranges for each key trace element by crystal type with relative standard deviation ranges in the new standards sets are presented in table 4. These can be compared to the targeted ranges of values in table 1. The homogeneity achieved in the crystal growth and naturally present in the Yogo sapphires are demonstrated by the low RSD values accompanying the concentration ranges for those trace elements used for calibration purposes. Looking at the range of values by trace element within a standard type underscores the significance of individually calibrating each piece from the same source.
Table 5 presents the final data with total combined uncertainty (includes all measurement uncertainties from all steps) for the first standards set imaged in figure 1. As mentioned, high uniformity of the trace elements within the crystals is necessary to improve accuracy and reproducibility while minimizing uncertainty. For each standard, we used SIMS to collect nine uniformly distributed spots across the entire surface. We found that the relative standard deviations representing the point-to-point variations in the calibrated levels of trace elements within our standards ranged from less than 0.5% to no more than 10%. The highest variation was in the diffused beryllium in the 02-1032 crystal pieces. The lowest variations were in the chromium in the synthetically grown crystals and in the iron in the Yogo sapphire slices (all 2% or less).
Preliminary Comparison of LA-ICP-MS Data Using the New Standards Versus NIST SRM 610 and 612 Glasses. To grasp the significance of matrix effects for key trace element reporting in corundum using NIST SRM 610 and 612 glasses, we compared values determined for one of our standards sets (set 7), first using our RSF values with SIMS and then with LA-ICP-MS using a different corundum standards set (set 1), and finally with LA-ICP-MS using NIST SRM 610 and 612 glasses separately. Additionally, we used the new corundum standards set 1 separately from NIST SRM 610 and 612 glasses to measure a series of other samples using LA-ICP-MS. We were not able to measure this set of samples on SIMS but plan to do so with additional samples in the future. As shown in table 6, using the new standards calibrated by SIMS in the LA-ICP-MS instrument provided agreement within less than 10% of SIMS for standards set 7. Looking at calibrating the LA-ICP-MS with NIST 610 and 612 as compared to the matrix-matched standards, trends in differences between the measured samples for each trace element can be seen. For Be, Ti, V, Fe, and Ga, NIST SRM 610 and 612 glasses produce values consistently lower than the corundum standards. For Mg, NIST SRM 610 and 612 glasses produce values higher than the corundum standards. For Cr, the trend is not so clear, but the differences are within less than 10% of each other.
Accurate Quantitative Reporting of Silicon in Sapphire Using SIMS. Using SIMS with an MRP of 3,000, we were able to completely separate the 27Al1H+ interference from 28Si+ (a mass scan around 28Si, collected on a sapphire with 22 ppma Si, at a chamber vacuum level of approximately 2 × 10–9 torr; see figure 5), allowing us to accurately quantify Si concentration in corundum. The quantitative reporting of silicon in sapphire is a problem for many of the LA-ICP-MS instruments used in gemological labs today, as they may not be capable of removing interferences and background from the isotope of interest, consequently yielding erroneously high results (Shen, 2010; Emmett et al., 2017). Using other techniques such as electron microprobe and XRF is not practical, as the detection limits for Si in corundum are orders of magnitude too high. LIBS with an intensified charge-coupled device (CCD) combined with a Czerny-Turner spectrometer might be more suitable than electron microprobe and XRF, but the limits of detection again for Si in corundum appear to be well above the 1 ppma range we are interested in.
Consider that the MRP for separating 28Si+ (mass of 27.97693 amu and 27Al1H+ (mass of 27.98936 amu) is 2,251. The MRP of the instrument for a particular mass is the ratio of the isotopic mass of interest to the peak width at half maximum. To determine the mass resolution required to separate a particular isotope from a close-in-mass interference, we take the mass of the isotope of interest and divide by the absolute value of the difference between that isotopic mass and the mass of the interference of concern. Here, for 28Si+ and 27Al1H+, the MRP is: 27.97693/(|27.97693 – 27.98936|) = 2,251. The MRP of quadrupole mass spectrometers used in many laboratories at atomic mass 28 is around 40 at normal resolution and tops out around 90 at high resolution. The required 2,251 MRP far exceeds the capability of these commercial “desktop” ICP-QMS used in a number of gemological laboratories. One could consider trying to determine the Si trace element concentration using the 29Si isotope (mass of 28.97649 amu), which has a much lower natural abundance (4.7% abundance vs. 92.2% abundance for 28Si). However, the signal from such a low-abundance trace element would be very difficult to quantify and distinguish from traces of Si that might be present due to quartz elements in the torch assembly. Even if one could correct for these possible problems, there are still close-in-mass interferences such as 28SiH and 29N-N (28.98475 and 29.00318 amu, respectively) that require finer MRP (3,508 and 1,085, respectively) than the quadrupole detector is capable of. We decided to test measuring trace levels of silicon in corundum using a magnetic sector field ICP-MS paired with a 193 nm LA unit. This work is still ongoing, but signal intensity for our spot size of choice is proving to be an issue for either 29Si or 28Si at the medium mass resolution option (4,000–5,000 MRP) to quantify the levels of silicon we found in our Yogo sapphire samples using SIMS (22–26 ppma).
For further context, the use of the high-purity “blank” sapphire containing essentially no trace elements allows the LA-ICP-MS operator to see at what masses erroneous quantities of trace elements may be detected. Where there is an inexplicable signal compared to what is expected for the matrix alone, one can assume the signal originates from sources including interferences from plasma products, other contaminants, and residual atmospheric components. In an alumina substrate, where Si concentration is very low, the dominant peak at mass 28 is 27Al1H+, which varies accordingly with the H contents (vacuum level) available during measurements.
To illustrate this further, we measured our ultra-high-purity synthetic sapphire blank using the laser ablation–inductively coupled plasma–quadrupole mass spectrometer (LA-ICP-QMS) calibrated with NIST SRM 610. We found erroneous and highly varying levels of both 28Si and 29Si in our high-purity blank of 1136 ± 333 ppma and 1062 ± 182 ppma, respectively (using SIMS, the 28Si level was below 1 ppma). This could help to explain in part the high levels of Si in corundum reported in previous literature using LA-ICP-MS and NIST SRM 610 or NIST SRM 612 standards (Peretti and Gunther, 2002; Peretti et al., 2003; Sutherland et al., 2015). After running a full mass scan from beryllium to uranium on the high-purity blank standard, also using NIST SRM 610 as a standard, we found that several trace elements beyond 28Si and 29Si would similarly be reported at erroneously high levels. These included 11B (around 50 ppmw), 43Ca (around 350 ppmw), 46Ti (around 2 ppmw), 56,57Fe (up to 18 ppmw), 67Zn (around 50 ppmw), 95Mo (around 0.4 ppmw, though this trace level may be contamination from the HEM crystal growth process), and 116,117,118,120Sn (around 0.5 to 2.5 ppmw).
CONCLUSIONS
The need for improved accuracy and efficiency led GIA to develop new corundum standards sets for its LA-ICP-MS instruments. These sets:
- Minimized the number of individual standards
- Incorporated a true “zero” to represent the matrix background
- Covered trace element levels that best represent the ranges found in natural ruby and sapphire of the key trace elements Mg, Ti, V, Cr, Fe, and Ga
- Contained individual pieces that were calibrated using a highly controlled process based on, to the best of our knowledge, the most accurate methods technologically available today
Additionally, we were able to calibrate silicon in corundum standards, and with our determined SIMS RSF value for silicon in sapphire we were able to accurately characterize trace levels of this significant trace element using the high mass resolving power of SIMS.
These new standards sets were tailored to ensure high reproducibility, minimal calibration time, and less chance of instrument signal drift with fewer individual measurements in the calibration cycle. The creation of these standards was a lengthy and complex endeavor, incorporating custom crystal growth, creation of ion implant standards, and utilization of RBS and SIMS measurement techniques. We consider the end product well worth the effort, as these new standards allow us to more accurately study corundum chemistry for the purposes of both basic research and geographic origin reporting.
Dr. Stone-Sundberg is a GIA research scientist and a technical editor of Gems & Gemology located in Portland, Oregon. Mr. Thomas is director and design engineering authority at Applied Materials in Hillsboro, Oregon. Mr. Sun is a staff gemologist at GIA in Carlsbad, California. Dr. Guan is laboratory director of the Microanalysis Center, Division of Geological & Planetary Sciences, at the California Institute of Technology in Pasadena, California. Mr. Cole is director of Scientific Materials in Bozeman, Montana. Dr. Equall is vice president of laser operations at FLIR. Dr. Emmett is director of Crystal Chemistry in Brush Prairie, Washington, and a consultant to GIA.

Dr. Stone-Sundberg is a GIA research scientist and a technical editor of Gems & Gemology located in Portland, Oregon. Mr. Thomas is director and design engineering authority at Applied Materials in Hillsboro, Oregon. Mr. Sun is a staff gemologist at GIA in Carlsbad, California. Dr. Guan is laboratory director of the Microanalysis Center, Division of Geological & Planetary Sciences, at the California Institute of Technology in Pasadena, California. Mr. Cole is director of Scientific Materials in Bozeman, Montana. Dr. Equall is vice president of laser operations at FLIR. Dr. Emmett is director of Crystal Chemistry in Brush Prairie, Washington, and a consultant to GIA.




