
ABSTRACT
Nephrites with similar hues usually have different ranges of saturation and tone, and color determination with the unaided eye can be problematic. The appearance of impurity ions such as Fe2+ influences the color of the material. Raman spectroscopy of 99 nephrite samples of different colors from northwestern China and Taiwan identified their main mineral as either tremolite or actinolite. When the Mg2+/(Mg2+ + Fe2+) ratio was 0.91 or higher, the mineral was tremolite; when the ratio was less than 0.91, the mineral was actinolite. The features in the M-OH stretching vibration region (3700–3600 cm–1) are important fingerprints of nephrite’s composition. Using the relative intensity ratio method, the authors calculated the Mg2+ and Fe2+ content and the ratio of Mg2+/(Mg2+ + Fe2+). In this study, the ratio was approximately 1 for white nephrite, 0.98–1 for light green nephrite, 0.90–0.98 for bluish green nephrite, and lower than 0.91 for dark green nephrite. The calculation of relative intensity ratios could provide a standard for nephrite color determination and even the critical evaluation of whiteness.
INTRODUCTION
Nephrite, an amphibole-rich rock, comes in a variety of colors (figure 1). As noted by Zhang (2006), these hues are classified by the Chinese as baiyu (white), qingbaiyu (white with a slight bluish, greenish, or grayish tint), qingyu (bluish green or greenish blue), biyu (green to dark green), huangyu (yellow), and tangyu (brown). Distinguishing the transitions between these color varieties is an important consideration in establishing a nephrite grading standard. Traders once determined the color solely by visual observation, but even a relatively accurate determination requires decades of experience. Major gemological laboratories still do not have a quantitative or semiquantitative boundary between colors. Since the value of fine white nephrite can be significantly higher than material in other hues, whiteness is the most important parameter in evaluating and classifying nephrite that ranges from white and gray to light green. The boundary between white and light grayish to bluish green nephrite is the hardest to identify, and establishing this difference is the focus of our study.
The amphibole group is comprised of double-chain silicates belonging to the monoclinic crystal system. The group’s general chemical formula is A0–1B2C5[T4O11]2 (OH, F, Cl)2. The components of the formula are described as A, B, C, T, and OH, with T corresponding to the tetrahedral sites in the unit cell. There are four kinds of sites occupied by cations (figure 2). These sites are C, a composite of five sites made up of two M1, two M2, and one M3 sites; and B, which is two M4 per formula unit. M1 and M3 sites are usually occupied by the Mg2+ and Fe2+ ions, and M2 sites by a trivalent or tetravalent form of cations with a smaller radius, such as Fe3+. M4 sites are occupied by Ca2+, Na+, and K+, while A sites are occupied by OH– and the remaining Na+ and K+ cations to balance the valence state. Cations and OH– that occupy M1, M3, and A sites form an ionic bond. Amphiboles are classified into four subgroups depending on the occupancy of the B sites: the Mg-Fe-Mn subgroup, the calcic subgroup, the Na-Ca subgroup, and the alkali subgroup (Bragg and Claringbull, 1965; Wang et al., 1982; Leake et al., 1997). Tremolite and actinolite are in the calcic amphibole subgroup.
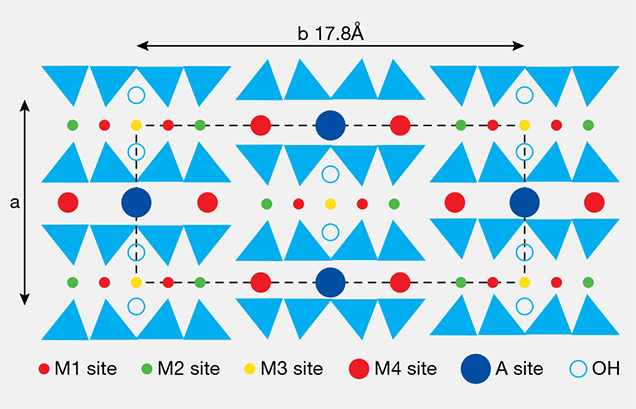
Nephrite is mainly composed of tremolite, Ca2Mg5[Si4O11]2(OH)2, and a variable amount of actinolite, Ca2 (Mg, Fe)5[Si4O11]2(OH)2. When the Mg2+/(Mg2+ + Fe2+) ratio is 0.91 or higher, the main mineral is tremolite; when the ratio is less than 0.91, it is actinolite (Bragg and Claringbull, 1965; Wang et al., 1982; Leake et al., 1997).
Many researchers exploring the role of Fe and Mg cations in the coordination of tremolite using different spectroscopic methods have pointed out that higher Fe2+ content generally produces a deeper green color in tremolite (Liu, 1988; Li and Wu, 1999; Guo and Han, 2002; Lu et al., 2007). On the basis of previous studies and our own Raman spectroscopic investigation of the relative intensity ratio of the spectral features related to M-OH bands, we propose a new method to calculate the relative content of Mg and Fe in the M1 and M3 sites. This method has the potential to define boundaries for nephrite color determination and may allow for evaluation of whiteness.
MATERIALS AND METHODS
Ninety-seven nephrite samples from China (Hetian in the Xinjiang Uygur Autonomous Region and Golmud in Qinghai Province) and two Taiwanese nephrites from Hualian were studied to form a nephrite jade grading standard. Of these, 15 samples showing a range of colors were selected for this study (table 1). One set of nephrite color standards was collected by the Xinjiang Uygur Autonomous Region Product Quality Supervision and Inspection Institute.

The nephrite samples were cut into wafers of different shapes. A Renishaw Raman 1000 spectrometer with a 514.5 nm laser was used at a power of 30–40 mW. To observe both the Si-O and M-OH stretching vibrations, Raman data were acquired with a spectral resolution of 4 cm–1 and a scanning time of 20 seconds in the 1200–100 cm–1 and 3700–3600 cm–1 ranges, respectively. Microprobe analyses were performed on a JEOL JXA-8800 electron microprobe analyzer at the Institute of Mineral Resources, Chinese Academy of Geological Sciences in Beijing. Analysis conditions were 20 kV accelerating voltage, 100 nA beam current, and a 5 μm beam.
RESULTS AND DISCUSSION
Raman Spectra. There are two regions of interest in nephrite’s Raman spectrum:
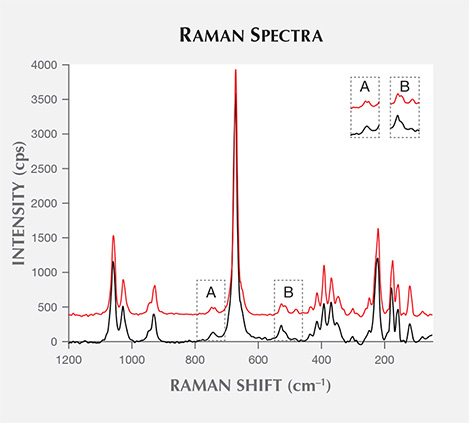
- The 1200–100 cm–1 region: Si-O vibrations are located in this range, where nephrite and other minor minerals can be identified. The main nephrite bands are shown in table 2 (Zou et al., 2002; Lu et al., 2007; Zhao and Gan, 2009). Our samples’ Raman spectra essentially match that of tremolite, indicating that this is the main mineral component of these nephrites (figure 3).
The main Raman peaks of tremolite-dominated nephrite are at 1062, 1031, 932, 741, 675, 530, 516, 437, 418, 396, 372, 353, 252, 225, 179, and 124 cm–1 (Zou et al., 2002; Wang et al., 2006).
With higher Fe content, the tremolite component gradually decreases while actinolite increases; at the same time, the Raman peaks shift accordingly (Lu et al., 2007). Some Raman peaks found in white and dark green nephrite (for instance, the peak near 741 cm–1, which is split into double peaks at 748 and 738 cm–1 in white nephrite and shifts to a single 743 cm–1 peak in dark green nephrite) shift about 1–5 cm–1, and their intensity diminishes or even disappears (again, see figure 3).
| TABLE 2. Main Raman bands of nephrite and theirassignments in the 1200–100 cm–1 region. | |
| Band Position (cm-1) | Assignment |
| 1062 | Si-O stretching vibration |
| 1031 | Si-O stretching vibration |
| 932 | Si-O stretching vibration |
| 741 | Symmetric Si-O-Si stretching vibration |
| 675 | Symmetric Si-O-Si stretching vibration |
| 530 | Si-O-Si and O-Si-O bending |
| 516 | Si-O-Si and O-Si-O bending |
| 437 | Si-O-Si and O-Si-O bending |
| 418 | Lattice vibrations |
| 396 | Lattice vibrations |
| 372 | Lattice vibrations |
| 353 | Lattice vibrations |
| 252 | Lattice vibrations |
| 225 | Lattice vibrations |
| 179 | Lattice vibrations |
| 124 | Lattice vibrations |
- The 3700–3600 cm–1 region: M-OH stretching vibrations are observed in this range. There are one to three bands, depending on the presence of Fe2+ in the M1 and M3 sites (Burns and Strens, 1966; Liu, 1988; Guo and Han, 2002; Lu et al., 2007). The number of peaks and their position also depend on the amount of Fe2+ occupying these two sites (table 3 and figure 4).
| TABLE 3. M-OH stretching bands of nephrite andcorresponding cations in the M1 and M3 sites. | |||||
| Fundamental peak maxima (cm–1) | 3675 | 3661 | 3645 | ||
| Cation Coordinating with OH– | M1 | Mg2+ | Mg2+ | Mg2+ | |
| M1 | Mg2+ | Mg2+ | Fe2+ | ||
| M3 | Mg2+ | Fe2+ | Fe2+ | ||
| Adapted from Burns and Strens (1966), Liu (1988), Guo and Han (2002),and Lu et al. (2007). | |||||

Raman spectra peak positions from M-OH stretching vibration in the nephrite samples are listed in table 4 and are summarized as follows:
- The white samples (H-01, H-02, H-03, and Q-01) showed a clear 3675 cm–1 peak but no 3661 or 3645 cm–1 peaks. This indicated very low Fe2+ content at the M1 and M3 positions, almost all of which are occupied by Mg2+ ions.
- The slightly greenish white or grayish white nephrite samples (H-04 and Q-02) showed peaks at 3675 and 3661 cm–1. The presence of the 3661 cm–1 peak indicated a larger amount of Fe2+ ions, but the 3661 cm–1 peak in these samples was very weak.
- The bluish green samples (H-05, Q-03, and Q-04) ranged from light bluish green to deep bluish green. Samples from Xinjiang had a lighter color than those from Qinghai; these lighter-colored samples showed Raman peaks at 3675 and 3661 cm–1 but for the most part no 3645 cm–1 peak. Both green samples from Qinghai exhibited a 3645 cm–1 peak. The 3661 and 3645 cm–1 peaks were more intense in the deep green nephrite, indicating that the green color varied with Fe2+ content (Lu et al., 2007).
- The dark green samples showed the 3675, 3661, and 3645 cm–1 peaks. This indicated a high Fe2+ content, which caused the dark green color.
- Peaks corresponding to the Fe3+ ion of M-OH stretching bands, such as those at 3631, 3651, and 3604 cm–1 (Guo and Han, 2002), did not appear in any of the samples. These samples probably contained little or no Fe3+, and Raman spectroscopy could not detect its presence.
| TABLE 4. Peaks from M-OH stretching vibration innephrite. | |
| Peak Position (cm-1) | Presence |
| 3675 | All Samples |
| 3661 | H-04, H-05, H-06, H-07, H-08, H-09, Q-02,Q-03, Q-04, TY-01, and TY-02 |
| 3645 | H-06, H-07, H-08, H-09, Q-03, Q-04, TY-01,and TY-02 |
The Relative Intensity Ratio Method. We used the relative intensity ratio of the 3675, 3661, and 3645 cm–1 peaks to calculate the relative content of Mg2+ and Fe2+ and the ratio of Mg2+/(Mg2++ Fe2+) in the M1 and M3 sites, and to classify the color of nephrite according to the Mg2+/(Mg2+ + Fe2+) ratio.
Raman spectroscopy is a powerful tool for quantitative analysis (e.g., Pelletier, 2003). In a study of titanomagnetites, Zinin et al. (2011) used the relative intensity of the Raman peaks to calculate the Ti concentration. In our study, the intensity of Raman spectra was related to sample composition. Based on the intensity of the spectral peaks, we could calculate the relative content of the corresponding cation. On the basis of previous studies, we used Raman spectroscopy to study the relative intensity ratio of the spectral peak in the M-OH bands and devised a new method to calculate the relative content of Mg and Fe in the M1 and M3 sites (figure 5). The color of each nephrite sample was differentiated according to this ratio.
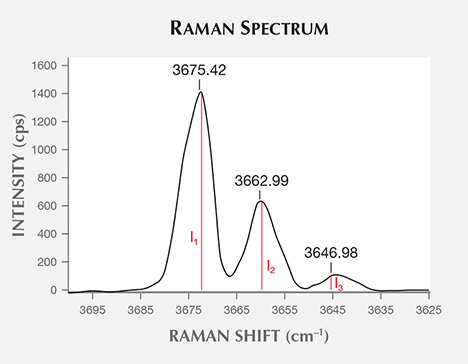
To calculate the relative Fe2+ and Mg2+ occupying the M1 and M3 sites, we set the relative intensity of the 3675 cm–1 peak at 1 (I1 = 1) and used this as a reference for calculation. The relative intensity of 3661 to 3675 cm–1 is I2 (I2/I1 = I2/1 = I2), while the relative intensity of 3645 to 3675 cm–1 is I3 (I3/I1 = I3/1 = I3). Based on the relative amount of Mg2+ and Fe2+ cations assigned to each peak position (again, see table 3), the relative amount of Mg2+ occupying M1 and/or M3 sites in a certain sample is calculated as I1 + 2I2/3 + I3/3, while the relative amount of Fe2+ is calculated as I2/3 + 2I3/3. We calculated the relative amounts of Mg2+ and Fe2+ in all samples and the Mg2+/(Mg2+ + Fe2+) ratio. The results are summarized in table 5.

According to table 5, the value of Mg2+/(Mg2+ + Fe2+) varies in samples by color. White nephrite has a value of approximately 1, light green nephrite 0.98–1, bluish green nephrite 0.90–0.98, and dark green nephrite below 0.91. The lower the value of the Mg2+/(Mg2++ Fe2+) ratio, the deeper the green color.
Comparison with Electronic Microprobe Data. To confirm the reliability of the relative intensity ratio method, we analyzed the chemical component of these samples using electron microprobe. The results are shown in table 6. According to the data, the Fe and Mg2+ content and the Mg2+/(Mg2+ + Fe) ratio can be calculated, as shown in table 7.>
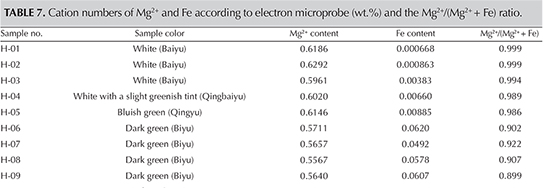
Comparing the Mg2+/(Mg2+ + Fe2+) ratio obtained using the Raman relative intensity method with electron microprobe results shows good agreement between the two methods, but there is some variance. The variance may be caused by several factors, starting with the different nature of the two methods. Second, the Fe content measured by electron microprobe includes both Fe3+ and Fe2+, while the spectral features reflect only Fe2+. In addition, the analyzing spot cannot be exactly the same, because the two instruments do not use the same magnification. Taking this into consideration, the difference of the ratio in these samples is less than 3%, an acceptable variance.
CONCLUSIONS
Raman spectroscopy can help distinguish the color varieties of nephrite (again, see figure 4 and table 5) using the relative ratio of Mg2+ to Fe2+ concentration. The Fe2+ concentration is lowest in white nephrite and highest in dark green nephrite. Since white nephrite (figure 6) tends to be highly valued, determining a boundary between white and light-colored nephrite is of importance to the trade. According to three relative peak intensity ratios in the M-OH stretching vibration bands, we can classify the color of nephrite to a certain degree. When the Raman spectrum only exhibits a 3675 cm–1 peak, the nephrite contains little Fe2+. If the peak is more intense, the nephrite is whiter. The presence of a 3661 cm–1 peak is associated with a green tone; it is this peak that separates white nephrite from slightly greenish white, bluish green, and dark green material. The Mg2+/(Mg2+ + Fe2+) ratio indicates the color of nephrite. The lower the ratio, the deeper the green color. When the relative intensity of the 3661 cm–1 peak is stronger, the nephrite has a darker color. The 3645 cm–1 peak only appears in dark green and bluish green nephrite. Thus, the method of relative peak intensity ratio of 3675, 3661, and 3645 cm–1 and the value of the relative intensity ratio are useful in evaluating nephrite color.

Ms. Feng is a senior engineer, Mr. Yong Zhang is an engineer, and Dr. Lu is chief researcher at the National Gems & Jewellery Technology Administrative Center (NGTC) in Beijing. Mr. Hui Zhang is the dean of Yunnan Land and Resources Vocational College in Kunming, China.

Ms. Feng is a senior engineer, Mr. Yong Zhang is an engineer, and Dr. Lu is chief researcher at the National Gems & Jewellery Technology Administrative Center (NGTC) in Beijing. Mr. Hui Zhang is the dean of Yunnan Land and Resources Vocational College in Kunming, China.




