
GIA’s Carlsbad laboratory examined a 4.21 ct faceted blue oval. Standard gemological testing determined that the stone was uniaxial negative and had a refractive index of nε = 1.759 and nω = 1.769, with a corresponding birefringence of 0.010. Hydrostatic specific gravity was measured as 4.00. The stone was inert to long-wave UV and showed medium chalky blue color to short-wave UV. These properties were consistent with heated sapphire, either natural or synthetic. Under magnification and diffused transmitted lighting, a blue color concentration was observed along the facet junctions on both the pavilion and crown facets, along with a few large blue halos on the table facet (figure 1, top left), which indicated that the stone had been titanium diffused (R.E. Kane et al., “The identification of blue diffusion-treated sapphire,” Summer 1990 G&G, pp. 115–133; Summer 2015 GNI, pp. 203–205). Under darkfield lighting, individual large spherical gas bubbles were present (figure 1, top right). Numerous clouds observed throughout much of the stone could have easily been mistaken for natural clouds. Some of them grouped to form natural-looking fingerprint-like inclusions (figure 1, bottom left). The stone’s internal characteristics indicated a melt-grown synthetic origin. However, when immersing in methylene iodide under crossed polarizers and viewing down the optic axis, no Plato lines were observed (see W. Plato, “Oriented lines in synthetic corundum,” Fall 1952 G&G, pp. 223–224). The absence of Plato lines does not rule out flame-fusion growth. The heat from diffusion treatment can reduce the visibility of Plato lines (S. Elen and E. Fritsch, “The separation of natural from synthetic colorless sapphire,” Spring 1999 G&G, pp. 30–41). More advanced testing is needed to reveal all the physical and chemical properties of this unique stone.
Under the short-wave radiation (a 20-nm-wide band centered at 225 nm) in a DiamondView device, the stone displayed a strong chalky blue fluorescence. Hughes and Emmett (2005) concluded that the chalky blue fluorescence was due to isolated Ti4+ ions, or Ti-Al vacancy pairs. The strength of the chalky fluorescence depends on the growth temperature and Ti4+ concentration relative to other impurities. In melt-grown corundum, the high growth temperature allows Ti4+ to pair with Al vacancies more easily than other charge compensating ions; this creates the chalky fluorescence. In contrast, Pairing of Ti4+ with other ions (usually Fe2+ or Mg2+) and the presence of Fe3+ ions prevent fluorescence. That could explain why the chalky fluorescence was not present where the blue diffusion color was concentrated (R. Hughes and J. Emmett, “Heat seeker: UV fluorescence as a gemological tool,” 2005, www.ruby-sapphire.com/heat_seeker_uv_fluorescence.htm).
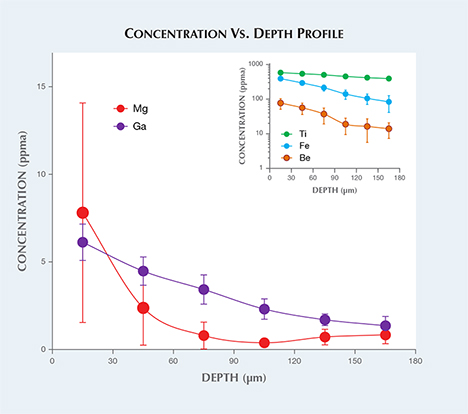
Laser ablation–inductively coupled plasma–mass spectrometry (LA-ICP-MS) analysis was performed on the girdle of the stone. Three laser ablation spots were drilled six times down into the stone. The concentration vs. depth profile is shown in figure 2. From the surface of the stone to around 180 µm depth, the Be concentration decreased from 76.8 to 13.9 ppma, Mg from 7.8 to 0.8 ppma, Ti from 580 to 391 ppma, Fe from 390 to 83.4 ppma, and Ga from 6.12 to 1.36 ppma. The results confirmed diffusion treatment, not only by Ti and Fe but also by Be, Mg, and Ga. The presence of Mo provided some indication of synthetic origin. In a GIA identification report, the stone was described as titanium and beryllium diffused. It is the first time GIA has examined a multi-elemental diffused and melt-grown synthetic sapphire.

GIA’s Carlsbad laboratory examined a 4.21 ct faceted blue oval. Standard gemological testing determined that the stone was uniaxial negative and had a refractive index of nε = 1.759 and nω = 1.769, with a corresponding birefringence of 0.010. Hydrostatic specific gravity was measured as 4.00. The stone was inert to long-wave UV and showed medium chalky blue color to short-wave UV. These properties were consistent with heated sapphire, either natural or synthetic. Under magnification and diffused transmitted lighting, a blue color concentration was observed along the facet junctions on both the pavilion and crown facets, along with a few large blue halos on the table facet (figure 1, top left), which indicated that the stone had been titanium diffused (R.E. Kane et al., “The identification of blue diffusion-treated sapphire,” Summer 1990 G&G, pp. 115–133; Summer 2015 GNI, pp. 203–205). Under darkfield lighting, individual large spherical gas bubbles were present (figure 1, top right). Numerous clouds observed throughout much of the stone could have easily been mistaken for natural clouds. Some of them grouped to form natural-looking fingerprint-like inclusions (figure 1, bottom left). The stone’s internal characteristics indicated a melt-grown synthetic origin. However, when immersing in methylene iodide under crossed polarizers and viewing down the optic axis, no Plato lines were observed (see W. Plato, “Oriented lines in synthetic corundum,” Fall 1952 G&G, pp. 223–224). The absence of Plato lines does not rule out flame-fusion growth. The heat from diffusion treatment can reduce the visibility of Plato lines (S. Elen and E. Fritsch, “The separation of natural from synthetic colorless sapphire,” Spring 1999 G&G, pp. 30–41). More advanced testing is needed to reveal all the physical and chemical properties of this unique stone.
Under the short-wave radiation (a 20-nm-wide band centered at 225 nm) in a DiamondView device, the stone displayed a strong chalky blue fluorescence. Hughes and Emmett (2005) concluded that the chalky blue fluorescence was due to isolated Ti4+ ions, or Ti-Al vacancy pairs. The strength of the chalky fluorescence depends on the growth temperature and Ti4+ concentration relative to other impurities. In melt-grown corundum, the high growth temperature allows Ti4+ to pair with Al vacancies more easily than other charge compensating ions; this creates the chalky fluorescence. In contrast, Pairing of Ti4+ with other ions (usually Fe2+ or Mg2+) and the presence of Fe3+ ions prevent fluorescence. That could explain why the chalky fluorescence was not present where the blue diffusion color was concentrated (R. Hughes and J. Emmett, “Heat seeker: UV fluorescence as a gemological tool,” 2005, www.ruby-sapphire.com/heat_seeker_uv_fluorescence.htm).
Laser ablation–inductively coupled plasma–mass spectrometry (LA-ICP-MS) analysis was performed on the girdle of the stone. Three laser ablation spots were drilled six times down into the stone. The concentration vs. depth profile is shown in figure 2. From the surface of the stone to around 180 µm depth, the Be concentration decreased from 76.8 to 13.9 ppma, Mg from 7.8 to 0.8 ppma, Ti from 580 to 391 ppma, Fe from 390 to 83.4 ppma, and Ga from 6.12 to 1.36 ppma. The results confirmed diffusion treatment, not only by Ti and Fe but also by Be, Mg, and Ga. The presence of Mo provided some indication of synthetic origin. In a GIA identification report, the stone was described as titanium and beryllium diffused. It is the first time GIA has examined a multi-elemental diffused and melt-grown synthetic sapphire.





