ABSTRACT
This article presents statistical data and distinctive features from several hundred faceted CVD-grown synthetic diamonds examined by GIA researchers from 2003 through June 2016. This study, the first comprehensive summary published on such a large number of gem-quality CVD synthetics, describes the reliable means of identifying them, with a focus on material currently marketed for jewelry use. Most CVD synthetic diamonds analyzed by GIA have been in the near-colorless or pink color ranges, with clarity grades comparable to those of their natural counterparts. Faceted CVD samples are generally 2 ct or less, though the sizes are increasing. They can be identified by their distinctive fluorescence pattern using the DiamondView imaging instrument, and by the detection of the silicon-vacancy defect using photoluminescence (PL) spectroscopy. Some visual gemological characteristics provide indicators, but not definitive proof, of CVD origin.
INTRODUCTION
Synthetic diamonds grown by the high-pressure, high-temperature (HPHT) process have been commercially available since the early 1990s. Perhaps several thousand faceted HPHT synthetics have been examined to date by gemological researchers; these specimens have been the subject of widespread coverage in Gems & Gemology and other trade publications. But gem-quality diamonds grown by chemical vapor deposition (CVD), which produces tabular crystals that are colorless or evenly colored, are of more recent manufacture. For these products, the range of observations and number of samples examined are more limited. While CVD growth technology has progressed significantly in recent years, with improvements in crystal size and quality, the use of these synthetics in the jewelry trade is still limited. The CVD material examined by GIA has generally contained minor amounts of microscopic inclusions. Therefore, the gemological identification of this material relies on the observation of weak, low-order, banded anomalous birefringence (i.e., “strain patterns”); ultraviolet fluorescence reactions with atypical colors, as seen with DiamondView imaging; and spectral features recorded mainly using photoluminescence (PL) spectroscopy. G&G has published a number of articles on the detection of CVD synthetic diamonds over the past decade.
This article presents statistical information and distinctive identification features based on a review of data from all the faceted CVD synthetic diamonds examined so far by GIA. Because no summary has been published on such a large number of CVD samples, we describe the most diagnostic means of identification, with an emphasis on the material currently being sold for jewelry use. We analyzed data gathered by GIA staff, principally at the New York and Carlsbad laboratories, on all CVD synthetic diamonds examined from 2003 through June 2016; additionally, we cite one CVD synthetic submitted after that date—a 5.19 ct “near-colorless” sample. Of this sample set, 60% were in the colorless to near-colorless to very light brown range (D–N) and 24% were in the “pink” color range (figure 1). The remaining 16% showed other colors, including gray, yellow, and brown. The samples were purchased by GIA on the market or from manufacturers, loaned or donated by manufacturers, or submitted to GIA for identification or grading reports. To the best of our knowledge, this data set of several hundred samples is representative of the gem-quality CVD material available in the trade.

Many of these samples have been described in detail in previous articles (Wang et al., 2003, 2007, 2010, 2012) and in G&G’s Lab Notes section (e.g., Wang and Moses, 2008; Wang and Moe, 2010; Ardon et al., 2013; Wang et al., 2013; Ardon and Wang, 2014; Moe et al., 2014). Whereas earlier studies examined small batches of CVD synthetic diamonds from specific manufacturers, which were likely to have been grown using a similar recipe, the goal of the present study is to investigate trends in the distinctive features seen among CVD synthetic diamonds from multiple sources over the past 13 years.
This article traces the evolution of gem-quality CVD technology from what we term the “pre-commercial” phase of 2003–2008. During this period, many advances occurred but the products were very much in the development stage in terms of crystal size and quality and were not widely available to the public. Since 2008, CVD synthetic diamonds of improved color and quality have gone into limited commercial production, though GIA’s laboratory still receives only a small quantity—both disclosed and undisclosed—from the trade. Of the CVD samples surveyed, 75% date from 2013 or later.
While the past decade has also seen advances in CVD technology aimed toward non-gemological applications such as quantum computing (Twitchen et al., 2016), we limit our observations to gem-quality CVD synthetics and the data set of material examined by GIA. Angus (2014) provides an excellent retrospective of the evolution of CVD growth prior to the “gem-quality” era covered here.
The synthetic diamonds discussed in this article are from only a few known manufacturers. In the past, GIA solicited samples from Apollo Diamond Inc. (which sold its technology to Scio Diamond Technology of Greenville, South Carolina, in 2011) and from the Gemesis Corporation of Bradenton, Florida (Wang et al., 2003, 2007, 2010, 2012). Some of the samples submitted to GIA’s laboratory are from other manufacturers, such as Washington Diamond or the Diamond Foundry, but not all the makers of our study samples are known, as many specimens submitted for grading reports came from third-party clients.
CVD SYNTHETIC DIAMOND GROWTH
The CVD process involves diamond growth at moderate temperatures (700–1300°C) but very low pressures of less than 1 atmosphere in a vacuum chamber (Angus and Hayman, 1988; Butler et al., 2009; Nad et al., 2015). In 1952, William G. Eversole at Union Carbide created tiny synthetic diamonds using the low-pressure, comparatively low-temperature method of CVD growth. Many of the early research efforts focused on (1) the “thermodynamic paradox” of growing diamond outside its stability zone and (2) exploring the chemical composition to suppress the simultaneous growth of graphite and the associated necessity of atomic hydrogen (Angus and Hayman, 1988; Angus, 2014). Single-crystal, gem-quality CVD synthetic diamond was once considered a “holy grail” within the research community. A half-century of investigation between CVD’s early beginnings in 1952 until it advanced sufficiently that it became a research focus at GIA in 2003 (Wang et al., 2003) made that once-ambitious goal a reality.
Square-shaped tabular single crystals up to approximately 13.5 ct can now be grown several at a time (Meng et al., 2012), at relatively fast growth rates up to 0.2 mm/hr (figure 2). Because growth occurs in a heated mixture of a hydrocarbon gas (such as methane, CH4) and hydrogen, the major impurity found in the resulting diamond crystals is hydrogen unless some other type of atom (such as nitrogen or silicon) is intentionally or unintentionally introduced into the gas mixture (Othman et al., 2014; Prieske and Vollertsen, 2016). Within the vacuum chamber, activation of the gas by an energy source (typically a microwave plasma) breaks apart the gas molecules to release carbon atoms. These atoms are drawn down toward the cooler substrate (typically flat, square-shaped seed plates) that consists of natural or, more typically, synthetic diamond. CVD synthetic diamond crystals are cubic in shape because they are often grown on a (100)-oriented diamond substrate: A different orientation of the substrate would yield a different crystal form. These “as-grown” tablets typically undergo laser cutting to remove the outer edge of the crystal, followed by standard gem polishing techniques (figure 3). Single-crystal diamond growth progresses, layer by layer on top of the seed plate, to form the tabular crystal. A slower growth process generally results in colorless crystals of very high purity and low defect content; however, the growth chemistry of many mass-produced CVD synthetics is manipulated by adding nitrogen or oxygen to the gas mixture to improve crystal quality (Liang et al., 2009), or they are treated at HPHT conditions afterward to remove any brown coloration. The tabular crystal shape generally limits the faceting styles used to manufacture gem-quality material. With the table facet of the cut stone as the upper surface of the crystal, the manufacturer has a small table-to-culet distance that may limit the choice of faceted shape.
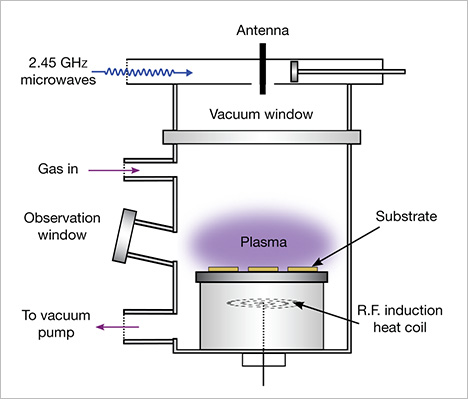

CVD synthetic diamonds have important non-jewelry uses in laser and electronic components, in optical windows, and as blades in cutting tools. These and other high-tech applications are the major driving force behind efforts to improve the growth process (Butler et al., 2009).
ANALYSIS OF EXISTING DATA
This study focuses on data obtained previously from two important categories of material, which appear to involve different post-growth color treatments. The first are the colorless to near-colorless to very light brown samples whose color grades fall within the D–N range of the GIA color scale; for brevity, these will be collectively referred to as “near-colorless.” Wang et al. (2012) concluded that these specimens were typically low-nitrogen type IIa synthetic diamonds and commonly subjected to HPHT annealing to remove a brownish color component (figure 1). The second group are type IIa “pink” samples (including brownish pink, pink, orangy pink, purplish pink, and a few red to pinkish orange); according to Wang et al. (2010), their colors are predominantly the result of post-growth annealing and irradiation of light brown starting material (again, see figure 1).
For these two groups, we will summarize quality factors, basic gemological properties, ultraviolet fluorescence reactions, and key spectral features based on information gathered when the samples were examined. In some instances, not all data types were collected on a particular CVD sample for various reasons: time constraints, the lack of certain instrument capabilities at the time, or the need to limit data collection to the client-requested grading service requirements. In the following discussion, therefore, the percentage of samples for which that data was collected will be clearly indicated. Nonetheless, the information from GIA’s internal records reported here represents the most substantial set of observations published to date on gem-quality CVD synthetic diamonds. Table 1 presents a brief timeline and summary of CVD synthetics examined by GIA.
Other observations, such as short-wave fluorescence, strain pattern between crossed polarizers, and near-infrared absorption spectroscopy, have not been routinely collected or reported (for exceptions, see Wang et al., 2003, 2007, 2010, 2012), and they are not discussed in detail here. Also, it should be noted that during the “commercial” period beginning in 2009, the instrumentation and data collection conditions used were consistent both over time and between GIA’s different laboratory locations.
We noted no significant differences in characteristics between the CVD samples sourced directly from the manufacturers and those submitted by GIA laboratory clients. The information presented here is strictly limited to material examined at GIA, which might not represent all of the goods available within the gem trade.
ANALYSIS OF QUALITY GRADING FACTORS
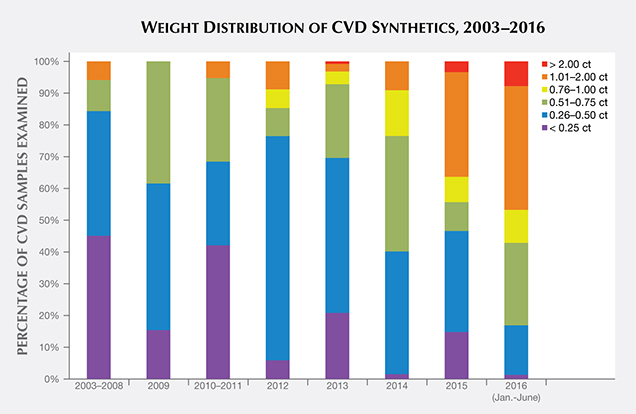
Carat Weight. Most of the faceted CVD synthetics examined by GIA fell in the 0.25–0.50 ct range (38%), with some larger than 1 ct (15%). In the pre-commercial era, samples below 0.25 ct were the most frequently examined. Between 2009 and 2013, the majority were in the 0.25–0.50 ct range. Since 2014, most have been larger than 0.50 ct (figure 4). This weight restriction is principally due to the tabular shape and the size of the as-grown crystals, which have influenced the choice of faceting styles and, in early material, the cut stone proportions.
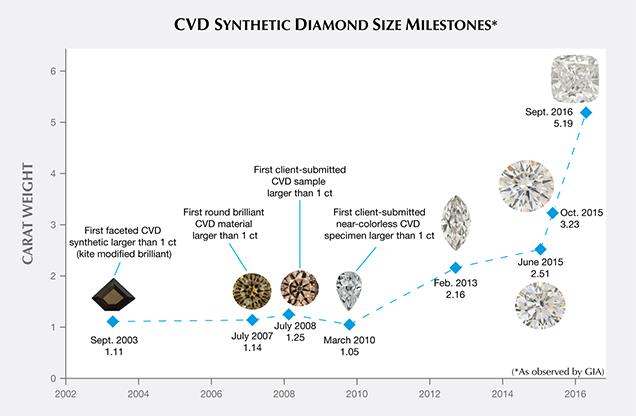
A few G&G Lab Notes have chronicled the increasing carat size of CVD material. Wang and Moe (2010) revealed the first client-submitted sample larger than 1 carat (1.05 ct, with G color and I1 clarity). Wang et al. (2013) reported on a 2.16 ct CVD synthetic (J/K color, SI2 clarity). In June 2015, the Carlsbad laboratory graded a 2.51 ct sample (H color, I2 clarity). In October 2015, the New York laboratory graded a 3.23 ct round brilliant (I color, VS2) clarity. In September 2016, GIA identified the largest CVD synthetic it has examined to date, a 5.19 ct cushion modified brilliant cut (J color, VS2 clarity; Law, 2016). Figure 5 shows some milestones in carat weight of CVD synthetics examined by GIA through the years.
Cut. The overwhelming majority of the faceted CVD synthetics were round brilliants (83%). Other styles included rectangle (3%), cut-cornered rectangle (3%), and emerald cut (4%). Many manufacturers seem to have chosen to facet the material as round brilliants because of the popularity of this style, even though “fancy” shapes would have retained slightly more weight from the original crystal.
The cut grades for the CVD round brilliants were comparable with their natural counterparts, with 85% judged as having an Excellent cut. The cut grade parameter of most interest was the depth percentage (the ratio of depth to average diameter); a low value would indicate the CVD synthetics were cut with lower than optimal pavilion or crown angles. As CVD synthetics are grown layer by layer, a shallow cut was once considered a possible indicator of CVD material (Wang et al., 2003). However, no discernible trend of this kind has been detected since 2007. Over these years, the CVD samples have demonstrated a consistent range of depth percentage, with the vast majority judged as Excellent for this parameter. Among those that fell outside of the Excellent range, most had higher depth percentages—an indicator that they were being cut with thicker girdles or larger crown or pavilion angles.
Color. Over the years, most CVD synthetics have been within the “near-colorless” category (figure 6): 67% in the G–J range, 21% in the D–F range, 10% in the K–N range, and only 1% in the split-grade range (O to Z on the D-to-Z scale). These percentages have not shifted dramatically over time.
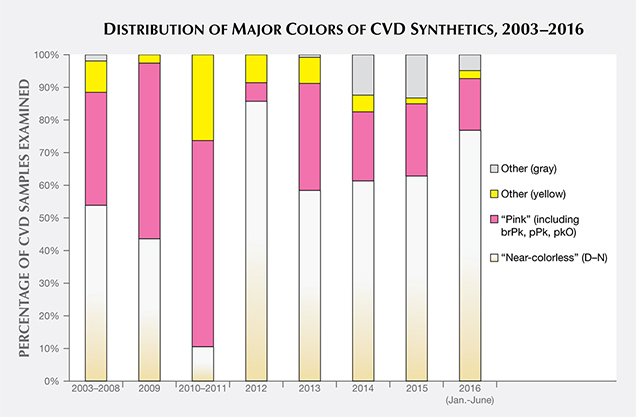
Wang et al. (2012) concluded that Gemesis products in the “near-colorless” range had been HPHT-treated after growth. Based on spectroscopic analysis, the conclusion applies to most CVD synthetics in this color range, regardless of manufacturer. The lower color grades on the D-to-Z scale are caused by either a yellow color from type Ib (i.e., single substitutional) nitrogen-related optical defects or a brown appearance due to extended defects (such as vacancy-related complexes). The post-growth treatment is presumably used to remove a brown or yellow color component so the material can obtain a better color grade.
The vast majority of the fancy-color CVD synthetic diamonds are within the pink range. Their hues include a variety of color descriptions such as pinkish brown, brownish pink, pink, purplish pink, and orangy pink, with one specimen described as red. Among the “pink” CVD synthetics, most fell in the pink (28%) to orangy pink (17%) to purplish pink (27%) range; these colors are due to multiple post-growth treatments, including HPHT annealing, irradiation, and low-temperature annealing (Wang et al., 2010). Except for a few samples (discussed in Wang et al., 2007; D’Haenens-Johansson et al., 2015), the “pink” color of the remaining CVD samples is attributed to nitrogen-vacancy (NV) centers (e.g., Wang et al., 2010). The colors are very similar to those of treated natural diamonds, treated HPHT synthetic diamonds, and rare type IIa Golconda-type untreated natural diamonds (table 2).
During the pre-commercial years and again more recently, GIA has graded CVD samples in other colors such as yellow and gray, and less frequently fancy brown and blue. These colors, which do not appear to be commercially important at present, are discussed briefly in box A.
BOX A: BROWN, YELLOW, BLUE, AND GRAY CVD SAMPLES
While CVD-grown diamond samples in colors other than pink have been examined by GIA over the years (figure A-1), these have not been submitted in significant quantities, and the individual colors do not appear to be commercially important.
Fancy-color brown CVD samples (some with orange or yellow modifiers) have been submitted by clients or manufacturers, particularly between 2003 and 2008. The brown color is attributed to non-diamond carbon inclusions and/or internal extended defects such as dislocations rather than the plastic deformation that causes brown coloration in natural diamonds (Wang et al., 2003).
Fancy-color yellow CVD samples (some with brown or orange modifiers) have been graded as well. The yellow color is ascribed to accidental or deliberate incorporation of nitrogen (e.g., Moe et al., 2014). These diamonds have nitrogen concentrations up to 12.5 ppm (e.g., figure A-1B and Ardon et al., 2013).
GIA has graded a few blue CVD synthetics in which the color is derived from irradiation (Ardon and Wang, 2014). In irradiated CVD-grown diamonds, as with irradiated natural diamonds, a strong GR1 absorption is created. This absorption has a sharp line at 741.2 nm and related absorption in the visible region; the latter imparts a green to blue color.
An interesting CVD creation is a pink-to-blue color change caused by massive quantities of intentional silicon doping (D’Haenens-Johansson et al., 2015). This is several orders of magnitude greater than the doping typically observed in CVD-grown diamonds and creates very strong SiV absorption. These heavily silicon-doped samples exhibit a color change from a stable light pink to an unstable blue. The pink-to-blue color change is caused by a charge-transfer effect between SiV– and SiV0 (D’Haenens-Johansson et al., 2015). The blue is created by strong absorption of the SiV0 center. This center exhibits a sharp absorption line at 946 nm within the NIR, together with absorption, which extends into the visible region and becomes less intense at shorter wavelengths. This color origin is similar to the cause of blue color in type IIb diamond in which the boron-related absorption starting in the IR shows decreasing intensity from the red end of the visible spectrum to the blue, causing the blue color. Pink is the stable color, and this is the color provided on the grading report.
More gray-colored samples have been submitted in recent years. Several of these had a depth of color that was nominally within the K–N range of the D-to-Z scale, but these received a fancy color grade since the dominant color was gray and not yellow or brown. It is likely that the manufacturer intended these to be near-colorless, falling on the D-to-Z scale.
The identification criteria for these colored CVD samples are similar to those for the near-colorless and pink material: a combination of spectroscopic and gemological evidence such as the presence of silicon and the growth features observed with the DiamondView.
Besides color, the other quality factors of carat weight, shape, and clarity are consistent with those previously described for the “near-colorless” and “pink” CVD synthetics.

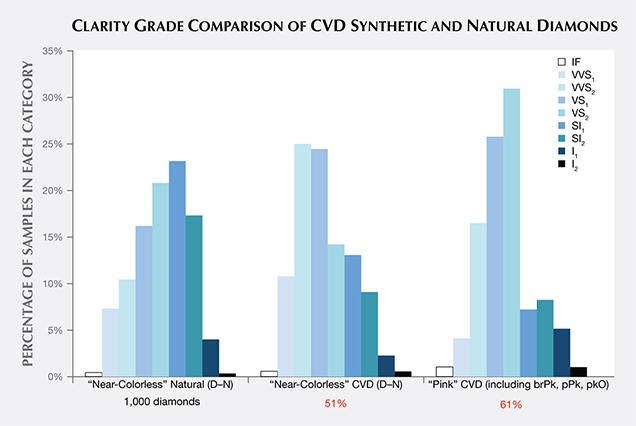
Clarity. The CVD samples span nearly the entire clarity scale, but most received grades in the VVS2/VS1 range. The clarity characteristics and grades for 1,000 randomly selected natural D–N diamonds submitted for grading reports were surveyed for comparison (figure 7). The median clarity of the CVD samples was slightly higher than for natural diamonds: VVS2/VS1 for “near-colorless” CVD, VS1/VS2 for “pink” CVD, and VS2/SI1 for near-colorless natural diamonds. This trend is not surprising, since the very controlled growth environment and growth chemistry of the CVD process leads to fewer inclusions and other visible clarity characteristics. As GIA generally examines only the material that CVD manufacturers decide is worthy of cutting, samples submitted to GIA are likely to reflect what is available within the trade but may not be representative of the manufacturing output.
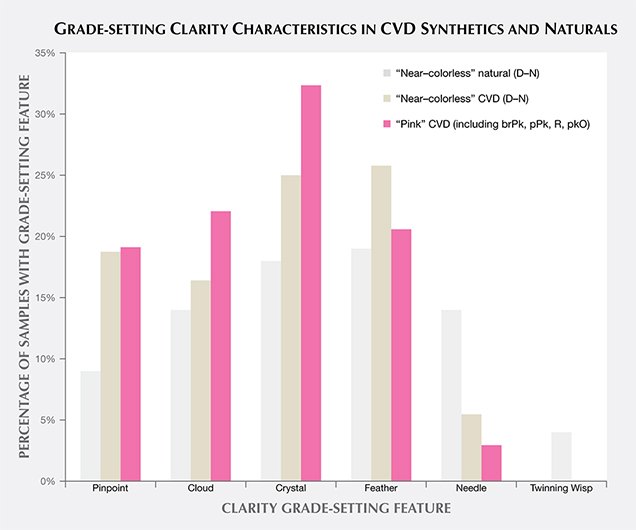
Clarity grading of diamonds often involves the determination of the “grade-setting” clarity feature. This is the characteristic that determines the clarity grade. For example, a diamond might have a large feather and a few sparse pinpoints; while both would be listed as inclusions, the feather would be considered the “grade setter.” A number of different grade-setting clarity characteristics were found in these CVD samples, as shown in figure 8. None of these characteristics would be diagnostic of this material for identification purposes, however. Certain manufacturers laser-inscribe a distinctive name or number on the girdle of their faceted goods as a recognition aid (figure 9).

The most common grade-setting inclusions for CVD synthetic diamonds were identified by GIA diamond graders as clouds, pinpoints, crystals, and needles; these inclusions were often mentioned in previous articles (Wang et al., 2003, 2007, 2010, and 2012). In most cases, these characteristics were black graphitic inclusions of various sizes (e.g., figure 10) or tiny pinpoints of non-diamond carbon that clustered together to form clouds. Wang et al. (2007, 2012) mentioned radial fractures surrounding larger inclusions; graders reported these as feathers if they reached the surface.

Despite their very different growth environments and histories, natural and CVD synthetic diamonds have clarity characteristics that are roughly comparable, at least in terms of size, number, location, nature, and relief. Therefore, GIA graders can use the same clarity terms for either. For example, a colorless foreign mineral in a natural diamond and a black inclusion of non-diamond carbon would both be described as a “crystal” by a grader (figure 10). A cloud is a cluster of pinpoints (tiny crystals that appear as specks at 10× magnification), while a crystal is solid mineral, larger than a pinpoint, contained within the diamond. A feather is an internal fracture visible at 10× magnification, and a needle is a thin crystal seen at 10× magnification. These clarity characteristics are annotated on internal GIA records as crystals, pinpoints, and clouds (among other designations) and are plotted accordingly for the client. On GIA synthetic diamond grading reports, they are not individually listed as crystals, clouds, or pinpoints, but collectively described under the umbrella term of “growth remnant.” Although similar terminology may be used, there are rare instances in which inclusions provide definitive evidence of diamond origin, such as metal platelets and garnet crystal inclusions.
It is interesting to note that twinning wisp inclusions, the grade-setting feature in 4% of the randomly selected natural diamonds, were not observed in any CVD synthetics. Twinning wisps are inclusions formed through an orientation change in the crystal structure during growth. Although twinning has been encountered in experimental CVD synthetics (Butler and Oleynik, 2008), such a change in the growth structure is unlikely to occur in current gem-quality CVD synthetics, as it would tend to lead to polycrystalline growth, something manufacturers wish to avoid.
ANALYSIS OF OTHER GEMOLOGICAL PROPERTIES
Long-Wave Fluorescence. The observation of ultraviolet fluorescence has been an important and practical means of distinguishing natural diamonds from HPHT-grown synthetics. The determination is based on differences in fluorescence colors, intensities, and patterns (and in some cases the occurrence of persistent phosphorescence among HPHT synthetics). UV fluorescence is also important for recognizing CVD-grown material.
The bar graphs in figure 11 illustrate the fluorescence reactions to long-wave ultraviolet radiation from a standard gemological UV lamp. Most of the “near-colorless” samples displayed no fluorescence intensity, while the “pink” samples tended to display stronger reactions. The distinguishing feature for both groups was the high incidence of orange and red fluorescence colors. Both of these colors, along with green, are extremely uncommon fluorescence reactions among natural, untreated diamonds.
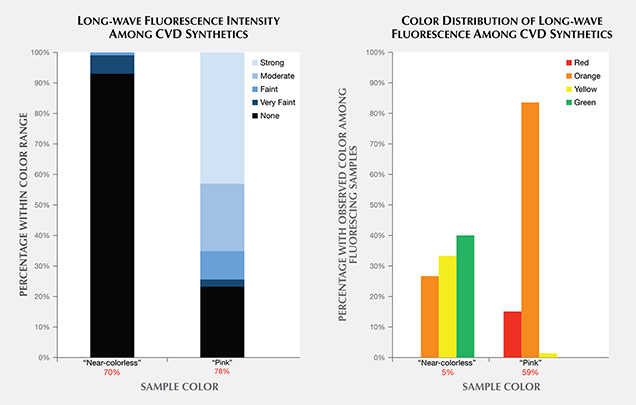
Near-Colorless Material. All CVD samples submitted to GIA for synthetic diamond grading reports are measured for the intensity of their fluorescence reaction to a long-wave UV source using a proprietary device. Only 7% of the “near-colorless” CVD synthetics had measurable (faint to very faint) fluorescence; the remainder had “none.” This is a far lower rate of fluorescent samples than observed among natural diamonds. Moses et al. (1997) reported that 35% of natural diamonds in the same “near-colorless” color range had detectable fluorescence, and that 99% of the fluorescence reactions were blue due to the N3 optical defect. By contrast, the small percentage of CVD synthetic diamonds with detectable fluorescence emitted orange, green, and yellow colors. Further investigation reveals that only the pre-commercial goods (2003–2008) showed orange fluorescence, while a few stones from 2010 and 2012 fluoresced yellow and green. The yellow to orange fluorescence is principally attributed to NV centers, and the green fluorescence to the H3 defect (Dobrinets et al., 2013).
None of the “near-colorless” CVD samples tested since 2013 had detectable long-wave fluorescence, indicating that the ever-evolving CVD process is no longer producing crystals that display observable fluorescence. It is unknown whether this trend represents a normal advancement in growth technology or a deliberate effort to remove a potentially identifying feature.
Pink Material. Due to the presence of NV defect centers acquired during post-growth treatment, the majority of “pink” CVD synthetics showed moderate to strong fluorescence, typically with orange or red colors. Only 23% had no reaction to long-wave UV. By contrast, 16% of the natural “pink” diamonds had no reaction to long-wave UV (King et al., 2002). Among those natural diamonds with observed fluorescence, the vast majority gave a blue reaction, while the remainder showed an orange color. Only a very small percentage (<1%) of natural pink diamonds derive their color from NV centers, and it is this small percentage that showed orange rather than blue fluorescence. Another trend among pink CVD synthetics in recent years is the increasing percentage of samples showing no fluorescence reaction. Before 2012, nearly all pink CVD synthetics tested had observable fluorescence; since 2013, fluorescence has been observed in only 68% of them. Other color origins for pink CVD synthetics include a ~520 nm band (Wang et al., 2007) or high silicon impurities (see box A); such material has also been submitted to the GIA labs.
DiamondView Imaging. For the past two decades, the DiamondView fluorescence imaging instrument has been a principal tool of major gemological laboratories. The design of the sample holder allows diamonds to be positioned and examined in various orientations, and the fluorescence reactions can be observed in real time. Because of the very short wavelength and intensity of the ultraviolet excitation source, fluorescence is created in diamonds at locations just beneath facet surfaces, producing a distinct reaction. Although many CVD synthetics showed no reaction to a standard long-wave UV light source, all diamonds—including all CVD synthetics—show some observable reaction to the high-intensity, high-energy UV source of the DiamondView.
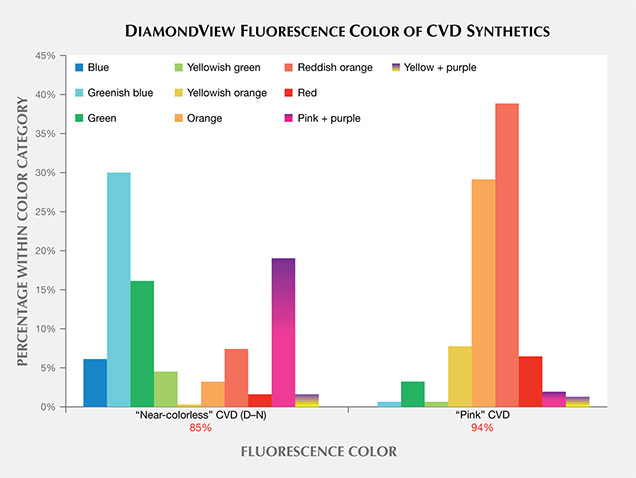
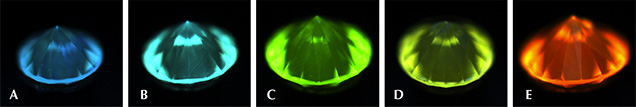
Differences in UV fluorescence colors and patterns in diamonds provide the basis for DiamondView analysis (Welbourn et al., 1996). Because of their unique growth environment and tabular crystal shape, CVD synthetic diamonds exhibit distinctive fluorescence reactions under the DiamondView. Figure 12 presents a histogram of the fluorescence colors observed in CVD samples using the DiamondView. The “near-colorless” samples on the left side of the figure exhibited a range of fluorescence colors, each due to a particular optical defect in the diamond lattice. By contrast, the “pink” samples displayed predominantly yellowish orange, orange, reddish orange, or red fluorescence reactions, which all appear to be the result of just the NV optical defect. Figure 13 shows the variety of colors and growth features that can be seen in CVD synthetic diamonds using the DiamondView.

Growth Features. As shown in figure 13, the fluorescence observed in the DiamondView is usually not distributed evenly. Thin striations of the luminescence are often visible (e.g., images D–H; see Martineau et al., 2004), especially through the pavilion facets. As mentioned earlier, CVD diamond growth takes place layer by layer on the top surface of the growing crystal. Minor or temporary disruptions in diamond formation can result in planes within the crystal where the fluorescing optical defects can become concentrated, and these are visible as striations.
In figure 13, images A and C illustrate a trend seen by GIA researchers in the last few years: a series of parallel bands, likely indicating interruptions during the CVD growth sequence. If a CVD specimen is grown for too long, single-crystal growth becomes polycrystalline growth (Nad et al., 2015). The only remedy is to stop the growth process, remove the crystal from the reactor, and polish away the non-gem polycrystalline diamond. The sample can then be returned to the reactor for the growth process to resume. The chemical composition within the gas phase and on the diamond surface is slightly different at start-up than when the reactor has reached a steady-state condition. Thus, the fluorescence is slightly different within this initial growth layer, as indicated by the evenly spaced luminescence bands (figure 13C). The interruption in growth is recorded as a distinct line in the fluorescence pattern. Such banded fluorescence patterns are not seen in natural diamonds in GIA’s labs or elsewhere (e.g., Mokuno et al., 2005; Nad et al., 2015).
Fluorescence Color in “Near-Colorless” CVD Synthetics. For the most part, the fluorescence colors seen in figure 13 originate from particular optical defects in the diamond lattice. The “near-colorless” CVD synthetic diamonds show a variety of fluorescence reactions due to the variety of growth and treatment histories for synthetic diamonds in this range—as-grown or HPHT-treated, grown slowly or quickly, grown with low accidental amounts of nitrogen or deliberately high amounts of silicon. The blue reaction, seen in about 5% of near-colorless material (figure 13C), is likely due to luminescence from bundles of structural dislocations (termed band A in the scientific literature; see Martineau et al., 2004). The greenish blue color is the most common observation among HPHT-treated CVD samples (figure 13D; Wang et al., 2012). The greenish fluorescence is ascribed to H3 centers (figure 13E; Wang et al., 2012).
The pink fluorescence alongside regions of purple mottling of dislocation bundles (figure 13B; see Wang et al., 2007) is observed in CVD synthetic diamonds in the “near-colorless” range and in a few pinks. These CVD synthetics showed the 596/597 nm doublet in their PL spectra. This indicates that the samples with the pink/purple DiamondView reaction are likely as-grown and were not subjected to additional treatment (see the “Photoluminescence Spectroscopy” section below).
Recently, several “near-colorless” CVD DiamondView images resembling the one in figure 13C (Eaton-Magaña, 2014) have displayed a pattern of dislocation networks and a deep blue color, which appears rather similar to some DiamondView images of natural diamond. The striations and distinctive colors, such as the pink/purple mottling in figure 13B or the light greenish blue in figure 13D, are not observed. The growth features in the deep blue fluorescing areas closely resemble the dislocation networks commonly associated with natural diamonds. This similarity illustrates the need for vigilance with all diamonds and confirms the importance of using more than one feature to identify a stone.
Fluorescence Color in “Pink” CVD Synthetics. As most pink CVD samples are treated after growth to create high concentrations of NV centers (the defect responsible for the pink color), it is not surprising that colors related to NV centers dominate the DiamondView fluorescence reactions. The yellowish orange to red reactions are due to the NV centers: the NV0 zero-phonon line (ZPL) at 575 nm and the NV– ZPL at 637 nm. The specific fluorescence color observed varies based on the relative concentrations of NV0 and NV– centers.
SPECTROSCOPIC PROPERTIES
With so few diagnostic visual gemological properties (namely the lack of color zoning or distinctive inclusions, but the presence of unusual fluorescence colors and banded strain patterns), spectroscopic techniques are essential for CVD detection in a gemological laboratory. These methods allow quick identification of diamonds as type IIa (such as using a DiamondCheck or DiamondSure instrument) and therefore as potential CVD synthetics. (Near-colorless type Ia diamond, which represents the vast majority of natural diamonds, still cannot be produced by the CVD technique.) Secondly, among the specimens identified as potential CVD synthetics, a combination of features in visible/near-infrared (Vis-NIR) absorption, Fourier-transform infrared absorption (FTIR), and PL spectroscopy will aid in definitive identification of CVD origin. Therefore, positive identification of CVD synthetics is best accomplished by a major gemological laboratory that maintains a database of information on known natural, treated, and synthetic diamonds and sees a sufficient quantity of goods to spot emerging trends.
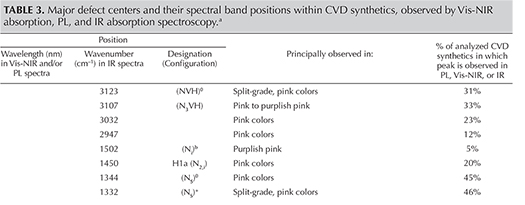
In the following discussion, the optical defects responsible for spectral features are indicated as either known or unknown to scientists. As an aid for the reader, table 3 summarizes the spectral features recorded by Vis-NIR, FTIR, and PL spectroscopy. We will focus solely on analyzing the presence or absence of the spectral feature. The relative intensity of the spectral peak will not be considered for this discussion because it is influenced by the size of the sample, the facet design and shape, and the path length of light travel within the material, among other factors.
In plotting these spectra, it was more illustrative to separate the “pinks” into brownish pink, pink, and purplish pink to show the progression in several spectroscopic properties with color and the different types of post-growth treatment. In other words, there were three categories: (1) HPHT decolorization treatment or no treatment for the “near-colorless” CVD synthetics, (2) no treatment or a growth method not commonly seen for the brownish pinks (e.g., Wang et al., 2007), and (3) HPHT annealing/irradiation/LPLT annealing for the pinks and purplish pinks.
Vis-NIR Spectroscopy. Figure 14 shows representative visible absorption spectra of a “near-colorless” and a “pink” CVD synthetic diamond. For the former, the lack of spectral features (and coloration) is typical of high-color type IIa diamonds. By contrast, the “pink” samples typically exhibit several sharp absorption bands, including several that are due to nitrogen-containing optical defects. Their Vis-NIR spectra appear quite similar to those of treated pink natural diamonds, illustrating the need for multiple measurement techniques and gemological observations to confirm an identification. Figure 15 shows how often these spectral features occur in the CVD synthetic diamonds from this study. Despite the fact that the samples are type II, nitrogen impurities are present in low concentrations (generally below the detection limit of FTIR absorption spectroscopy, so that the sample is still considered a type IIa diamond), and their visible spectra display features that originate from nitrogen-containing optical defects. These features indicate a small amount of nitrogen in the gas mixture during diamond synthesis.
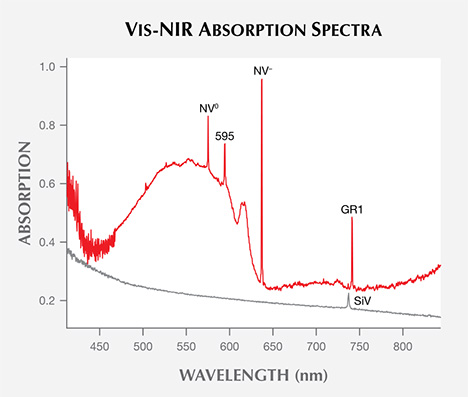
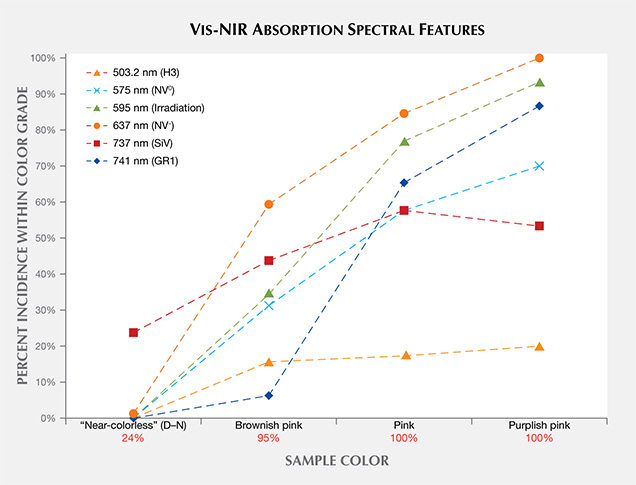
As expected, Vis-NIR spectra for “near-colorless” CVD synthetic diamonds are nearly featureless. Only a small percentage of these samples show the silicon peak at 737 nm, and the silicon-vacancy (SiV) absorbance in these samples is too weak to noticeably affect their color. As color progresses from the “near-colorless” to the “pink” range, there is a much higher incidence of nitrogen-related spectral features, including NV– (at 637 nm), NV0 (575 nm), and H3 (503.2 nm).
The brownish pink CVD samples were occasionally as-grown, with high concentrations of silicon, or the pink color was derived from an absorption band centered at approximately 520 nm (Wang et al., 2007, Khan et al., 2009, Khan et al., 2010) rather than high concentrations of NV centers at 575 and 637 nm.
The post-growth treatment that creates the pink and purplish pink colors usually involves a combination of irradiation and annealing. As expected, the irradiation-related optical defects (the GR1 and 595 nm centers) and related absorption features are significantly higher for these samples.
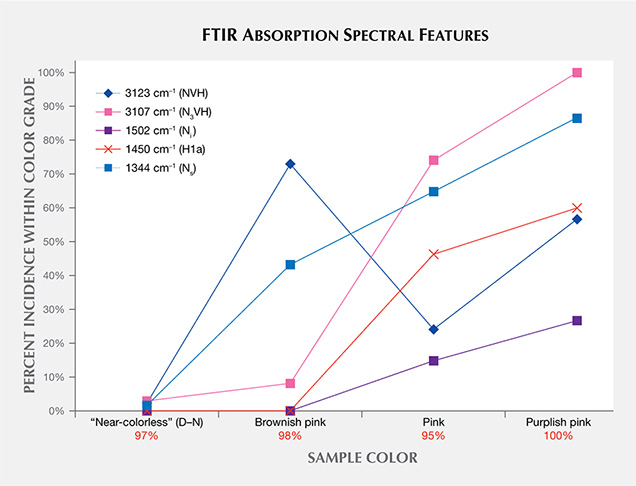
Infrared (IR) Absorption Spectroscopy. IR absorption spectroscopy is most useful in separating type IIa “near-colorless” diamonds—and therefore CVD and HPHT synthetics—from type Ia, which represent the vast majority of natural diamonds. (This is the basis of GIA’s DiamondCheck instrument.) In addition, several features in the IR spectra indicate CVD origin or provide an indication of post-growth color treatment. Figure 16 shows representative IR absorption spectra of two CVD samples: as-grown “near-colorless” and treated “pink.” With post-growth treatment, the 3123 cm–1 peak becomes dramatically weaker, while both the 3107 cm–1 peak and the H1a peak at 1450 cm–1 are created. As shown in the figure, the detected features are generally quite weak.
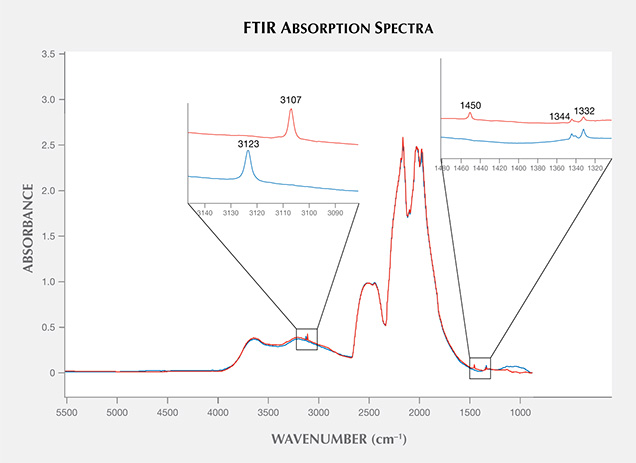
Nitrogen concentrations at the atomic-parts-per-million (ppma) level of detection may be calculated using IR spectra. Nitrogen concentrations were determined using a customized algorithm derived at the Diamond Trading Company (DTC) Research Centre in Maidenhead, UK (from Kiflawi et al., 1994; Boyd et al., 1994, 1995). Most “near-colorless” CVD-grown diamonds had no detectable nitrogen aggregates in their IR spectra, indicating nitrogen concentrations of less than 1 ppm. The treated pink, as-grown brown-pink, and O-to-Z samples (which are all nominally type IIa) showed weak absorption at 1344 and 1332 cm–1, indicating the presence of 1 to 5 ppm of substitutional nitrogen.
Figure 17 shows the incidence of particular features seen in the mid-IR spectra of the CVD samples. Despite the fact that this CVD material is type IIa (indicating a very low nitrogen concentration), all of the nonintrinsic peaks included in figure 17 originate from various nitrogen-containing optical defects.
Photoluminescence (PL) Spectroscopy. PL spectroscopy is a very sensitive analytical technique that can detect optical defects at much lower concentrations than absorption spectroscopy. Therefore, the PL method (where spectral features are excited by incident laser light) is capable of detecting optical centers in diamond when Vis-NIR absorption is not. PL is a vital technique for gemological laboratories in determining diamond origin and color origin, and CVD synthetics are no exception. Figure 18 shows representative PL spectra for two near-colorless CVD samples: one that did not undergo post-growth color treatment (as evidenced by the 596/597 nm doublet) and one that was HPHT annealed, which had a much lower concentration of NV centers and no 596/597 nm doublet. Both show a very small but detectable peak related to the SiV– center. The 596/597 nm doublet has never been reported in natural diamond, HPHT-treated CVD synthetics, or CVD synthetics that have undergone multiple treatment steps. It is considered a reliable indicator that a CVD specimen has not undergone HPHT treatment (Wang et al., 2010, 2012).
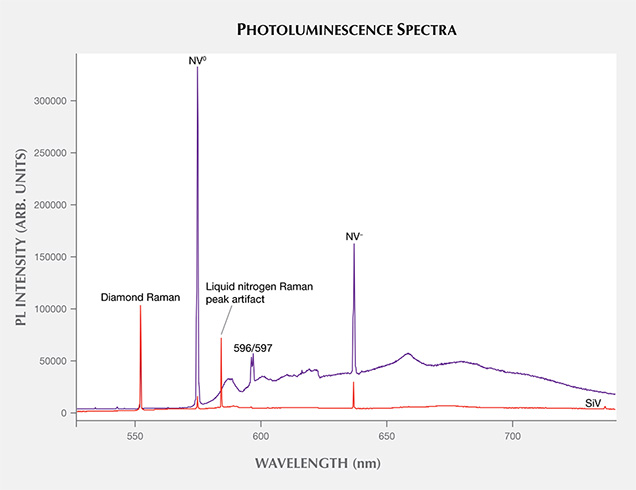
The SiV– doublet at 736.6 and 736.9 nm is a strong indicator of CVD growth; the center is seen very rarely in natural diamonds and occasionally in HPHT synthetics (Breeding and Wang, 2008). Other features, such as the presence of the 596/597 nm doublet, indicate that the CVD sample did not undergo any color treatment, while the H2 center is an indicator of post-growth treatment (figure 19). With synthetic diamonds, however, the jewelry trade makes no distinction in market value between as-grown and treated products.
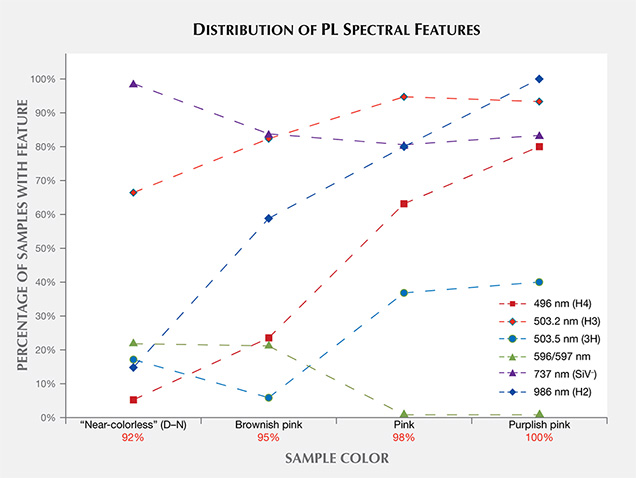
Figure 19 shows the incidence of selected features in the PL spectra based on sample color. In this figure, the percentage of CVD-grown diamonds with the 596/597 nm doublet decreases as the color progresses from “near-colorless” to brownish pink to pink. This also provides an estimate of the percentage that have not undergone post-growth treatment. Among “near-colorless” samples, 30% have the 596/597 nm doublet, indicating that the remaining 70% were likely HPHT-annealed. The pink samples showed a higher percentage of H2, H3, and H4 centers due to high-temperature heating during post-growth treatment for these color ranges.
CRITERIA FOR THE GEMOLOGIST
There are no reliable visual factors to help identify potential CVD synthetics. The size of the cut stone is not diagnostic: Most CVD synthetic diamonds are below 0.75 ct, and (except for one recent sample) all those surveyed by GIA have weighed less than 4 ct. The color appearance and the cause of the pink color are not diagnostic for the majority, though the pink color in CVD synthetics is usually more saturated than in natural pink diamonds.
There are several distinctive features gemologists may use to recognize in order to identify potential CVD synthetics, though none should be relied upon as the sole means of identification.
- Orange-to-red fluorescence reaction to a standard UV lamp in pink specimens derives from NV centers, indicating that the diamond is either a rare Golconda-type natural diamond, a treated natural diamond, or an HPHT or CVD synthetic (table 2). Although the vast majority of “near-colorless” CVD synthetics do not show observable long-wave fluorescence, some older material shows fluorescence colors (such as green) that are quite rare among natural colorless diamonds. In a gemological laboratory, the presence of striated fluorescence corresponding to multiple growth layers in DiamondView images provides additional confirmation of CVD growth, but its absence does not eliminate the possibility of CVD origin.
- CVD samples span the clarity scale, but those observed by GIA’s laboratory trend toward higher clarity grades. The small inclusions most often encountered are clouds, pinpoints, and black “crystals.” Similar clarity features are found in both natural and CVD synthetics. These inclusions are generally not diagnostic for identification purposes, despite the very different diamond formation environments.
- Type II diamonds—samples that are UV transparent or show no detectable nitrogen in their FTIR absorption spectra—should always be sent to a laboratory for testing.
- Major CVD manufacturers inscribe the synthetic diamond’s origin on the girdle (e.g., “Lab Grown”). But note that the lack of an inscription does not necessarily indicate a natural stone.
- A strain pattern viewed in the polariscope or a microscope configured with crossed polarizing filters (e.g., figure 20) can help distinguish CVD synthetics from HPHT synthetics, as the latter show extremely low levels of strain (e.g., figure 7 in D’Haenens-Johansson et al., 2015b).

Gem-testing laboratories need to maintain a full complement of gemological, imaging, and spectroscopy instrumentation, including a DiamondView imaging microscope, an FTIR absorption spectrometer, and a photoluminescence spectrometer equipped with several lasers to distinguish diamond origin and treatments. Additionally, they must have access to CVD specimens that are representative of products available in the trade and maintain a database of information on their properties. They must be constantly on the lookout for emerging trends that deviate from standard detection criteria for natural, treated, or synthetic diamonds. Gem laboratories must also operate under the assumption that today’s reliable criteria may not be applicable in the future and find alternative methods to ensure that identification keeps pace with the manufacturers’ technology.
FUTURE
Melee. In recent years, several gemological laboratories (including GIA’s) have established melee sorting services for colorless to near-colorless diamonds to separate natural stones from those that are potentially treated or synthetic. Therefore, both colored melee (through grading reports) and colorless melee (through sorting) can be screened for synthetics. Yet melee-size CVD synthetic diamonds have not been routinely submitted to GIA for examination. Based on our own observations and discussions with manufacturers, there is little evidence of CVD-grown melee in the marketplace. We have recently examined some CVD melee (e.g., figure 21) and anticipate that melee-size CVD material will become more available in the future (Poon et al., 2016). There have been reports that CVD melee is being manufactured in India, and this supply might become more prevalent in the coming years (W. Wang, pers. comm., 2016). HPHT-grown melee will likely remain more common than CVD-grown material, since yellow samples that can be fashioned as melee are easy to produce by this method. Although colorless natural melee is more abundant, HPHT-grown synthetic diamonds are currently in limited supply.
Without our own data or any published information, it is difficult to know the extent of HPHT and CVD synthetic diamond melee in the marketplace. Because of the availability of natural colorless melee, pink CVD melee is more likely to be produced (e.g., Kitawaki et al., 2010). If this is the case, pink CVD melee are not being submitted to GIA, which accepts colored diamonds of melee size for origin reports.
Outlook. Several features seen in early CVD synthetics have become less relevant in recent years. CVD-specific peaks such as 3123 cm–1 in IR absorption spectra and the 596/597 nm doublet in PL spectra were common in early products. Most CVD material today has undergone at least one round of post-growth treatment (either HPHT annealing or a combination of irradiation and annealing), so these early features are rarely observed. Likewise, it is possible that today’s criteria will no longer be applicable in a few years. Growth techniques are rapidly progressing due to research by CVD manufacturers for gem purposes and high-tech applications.
At present, the reliable identification criteria for undisclosed client-submitted samples have been established by examining CVD synthetics from known manufacturers: for instance, the presence of the SiV peak in Vis-NIR absorption or PL spectroscopy, and synthetic growth features as observed by DiamondView luminescence imaging.
While quality factors such as color, the cut grade of round brilliants, and clarity have been fairly consistent in CVD synthetics over the last several years, carat weight has risen. The number of samples submitted to GIA dwindled during 2010–2011, when Apollo Diamond Inc. was acquired by Scio, but has increased dramatically since then as Scio and other manufacturers have ramped up their production. Gemesis added CVD synthetics to their offerings in 2012 (Wang et al., 2012), and more companies have starting selling their own manufactured products.
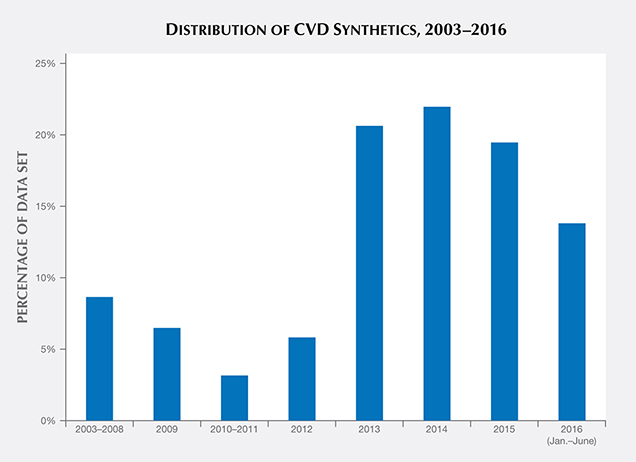
It is likely that the range of CVD synthetic colors will continue to expand and the sizes will increase. In September 2016, GIA’s Hong Kong laboratory graded a 5.19 ct J-color CVD synthetic—the largest submitted to GIA thus far (Law, 2016). Also, there have been unique CVD synthetics with interesting optical characteristics, such as samples with a reversible color change between pink and blue due to a charge-transfer effect caused by high amounts of incorporated silicon (D’Haenens-Johansson et al., 2015; see also box A). We also expect the quantity of CVD synthetics submitted for grading reports to increase in the coming years. Figure 22 shows the percentage of CVD synthetics within the data set from this study divided by year. There was a marked increase in CVD samples examined by GIA between 2012 and 2013, and this trend should continue to increase as new CVD diamond manufacturers (e.g., Bates, 2015) release their products.
CONCLUSIONS
Today the CVD process is used to produce high-color (or fancy color) and high-clarity type II synthetic diamonds up to several carats in size that are likely to be visually indistinguishable from polished natural diamonds. These CVD synthetics may display ultraviolet fluorescence reactions and inclusion features that would be unusual within natural diamonds. Additionally, the results described in this article illustrate how the samples all exhibited spectroscopic features that were quite different from those occurring in similar natural gem diamonds. Based on the use of analytical instruments and access to our large database of gemological information, the synthetic diamonds examined to date by GIA’s laboratory can be readily identified.
Dr. Eaton-Magaña is a research scientist, and Dr. Shigley is a distinguished research fellow, at GIA in Carlsbad, California.
Dr. Eaton-Magaña is a research scientist, and Dr. Shigley is a distinguished research fellow, at GIA in Carlsbad, California.




