Phosphorescence of Synthetic Sapphire

Phosphorescent features in synthetic colorless sapphires have previously been reported in G&G (Fall 2013 GNI, pp. 182–183). A 1.14 ct colorless sample was recently submitted to the New York lab for identification (figure 1, left). The gemological properties determined the stone was corundum. The specimen was free of inclusions, with no detectable gas bubbles or curved striae. Quantitative chemical analysis, showing a lack of gallium as well as iron levels below the detection limit, indicated that the sample was synthetic. The material was also transparent to short-wave ultraviolet radiation, whereas natural colorless sapphires do not transmit short-wave UV light due to the quenching effect from trace impurities (S. Elen and E. Fritsch, “The separation of natural from synthetic colorless sapphire,” Spring 1999 G&G, pp. 30–41). This is the same test used to effectively separate type I and II diamonds; nitrogen impurities in diamonds are strong absorbers of light in the short-wave UV range between 225–320 nm (C. M. Breeding, “The ‘type’ classification system of diamonds and its importance in gemology,” Summer 2009 G&G, pp. 96–111). The UV-visible spectrum shown in figure 2 also illustrates the transparency with the absorption edge at approximately 216 nm, whereas natural near-colorless sapphires typically have an absorption edge around 300 nm.
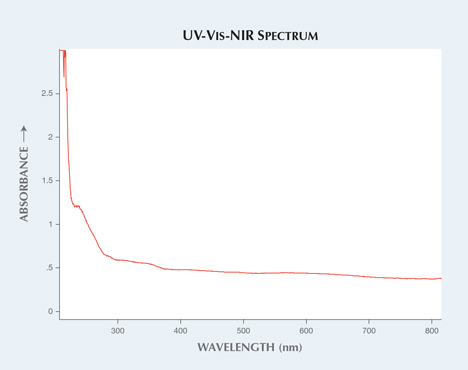
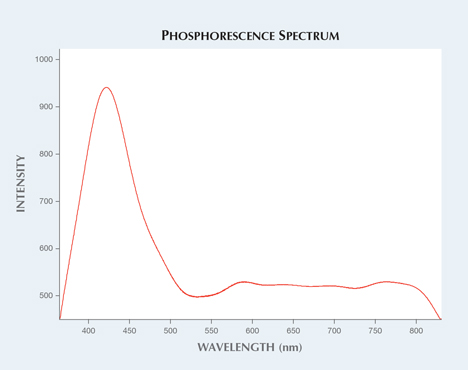
Figure 3 illustrates the phosphorescence band centered at approximately 424 nm, along with other minor decay bands observed in the visible spectrum that mix together to produce the blue phosphorescence observed in figure 1 (right). The phosphorescence lasted about 25 seconds, until the excited electrons dissipated. The only trace elements detected with LA-ICP-MS were Ti (21 ppma average), and Cr, Mg, and Ca (below 5 ppma). Chalky blue fluorescence in sapphires is believed to be due to Ti4+ charge-transfer transition of isolated Ti4+ ions or Ti-Al vacancy pairs. The fluorescence peak is usually between 410 and 420 nm, but shifts to higher wavelengths as Ti concentrations increase (B. D. Evans, “Ubiquitous blue luminescence from undoped synthetic sapphire,” Journal of Luminescence, Vol. 60–61, 1994, pp. 620–626; W.C. Wong et al., “Charge-exchange processes in titanium-doped sapphire crystals. I. Charge-exchange energies and titanium-bound exitons,” Physical Review B, Vol. 61, No. 9, 1995, pp. 5682–5698). The phosphorescence in synthetic colorless sapphires could be related to this property, with the addition of the decay route including forbidden energy transition states. Phosphorescence in synthetic colorless sapphires is still not well understood, and additional research is needed to verify the origin of this phenomenon.