Cultured Pearls from Lake Kasumigaura: Production and Gemological Characteristics

ABSTRACT
Although today’s global freshwater cultured pearl market is mostly dominated by Chinese products, Japanese freshwater pearl cultivation started in 1935. Lake Biwa in Shiga Prefecture supplied pearls in a wide variety of colors to the domestic and international markets until 1982. Due to water pollution and the depletion of Hyriopsis schlegelii, some of the pearl farms relocated to Lake Kasumigaura in Ibaraki Prefecture starting in 1962. Today the annual production of large nucleated cultured pearls at Lake Kasumigaura is below 40 kg, a small portion of which are supplied to the international market. This report investigates “Kasumiga pearl” culturing and describes the quality and production volume. UV-visible spectroscopy, fluorescence testing, and chemical analysis were performed on pearls in six different colors collected from the Hyriopsis schlegelii × Hyriopsis cumingii hybrid mollusk farmed at Kasumigaura.
INTRODUCTION
Since the initial success of culturing round pearls using the akoya oyster in the early twentieth century, there has been a consumer demand for pearls in a variety of colors, shapes, and sizes. Freshwater pearl culturing in Japan began in 1935, with commercial production of nucleated (beaded) pearls at Lake Biwa, Japan’s largest lake. These were produced using Hyriopsis schlegelii mollusks grown in Lake Biwa. After World War II, cultivation was shifted from beaded pearls to non-nucleated pearls. The annual production of freshwater pearls in Japan peaked at seven tons per year between 1970 and 1980 (“Statistics of fishery and cultivation,” 2000), at the same time Chinese freshwater pearls were entering the market.
In 1962, with natural resources of Hyriopsis schlegelii in Lake Biwa facing depletion, some Japanese freshwater pearl farmers started moving to the Lake Kasumigaura region in Ibaraki Prefecture to set up a new cultivation base (figure 1). With the need for stronger pearl-producing oysters, Hyriopsis schlegelii and Hyriopsis cumingii were crossbred to produce a Hyriopsis hybrid (figure 2). This oyster can grow larger and more lustrous nucleated cultured pearls in various colors (figure 3). Current annual production of freshwater cultured pearls in Lake Kasumigaura—Japan’s second-largest lake—remains low, at less than 40 kg, but these “Kasumiga pearls” are quite popular among buyers from Europe and the United States. This report examines current Japanese freshwater pearl culturing and the characteristics of the pearls produced at Lake Kasumigaura.


HISTORY OF FRESHWATER PEARL CULTURING IN JAPAN
Lake Biwa. Freshwater pearl culturing dates as far back as the thirteenth century, when pearls bearing Buddhist images and hemispherical pearls were produced from Cristaria plicata in China (Ward, 1985). In Japan, freshwater pearl culturing started during the Meiji era (1904–1912) with Tatsuhei Mise using Cristaria plicata at Lake Kasumigaura followed by Tokujiro Koshida’s experiments with Margaritifera laevis at Chitose River in Hokkaido, but both trials ended in failure. Masao Fujita tried a number of experiments in and around Lake Biwa and succeeded in commercial freshwater pearl cultivation using Hyriopsis schlegelii in 1935. His breakthrough was interrupted by World War II, and with the resumption of culturing operations came a shift from nucleated to non-nucleated pearls, which eventually formed the foundation for modern freshwater pearl cultivation (Wada, 1974; Akamatsu et al., 2001).
Due to war, the first year of recorded production at Lake Biwa was 1955. In that year, the Japan Ministry of Agriculture, Forestry, and Fisheries reported 0.1 tons of freshwater cultured pearls. This figure increased to between six and seven tons during the 1970s, with exports going mainly to the Middle East and India (Toyama, 1991). More than 99% consisted of non-nucleated pearls, and less than 1% were beaded pearls. Farmers in Lake Biwa successfully produced different shapes (figure 4) such as “dragon,” cross, triangle, long stick, or triplet, in addition to conventional round or baroque shapes (Wada, 1999). In addition, large nucleated pearls over 10 mm in diameter were cultured in very limited quantities. A wide variety of colors such as pink, purple, orange, brown, and blue, which were not possible by the traditional culturing method using Hyriopsis schlegelii, were achieved by grafting a piece of Anodonta calipygos kobelt mantle tissue into Hyriopsis schlegelii.

In the 1970s, freshwater non-nucleated pearl culturing began along the Chang Jiang River in central mainland China, utilizing its favorable natural environment and abundant freshwater mussel resources. About 13 tons were exported to Japan in 1980, based on statistics published by the Japanese Ministry of Finance. This greatly surpassed the total amount being produced in Japan (about seven tons per year), and Chinese annual production volume has grown larger every year, reportedly surpassing 1,000 tons (Akamatsu et al., 2001).
Meanwhile, pearl culturing at Lake Biwa went into steep decline. Natural resources of Hyriopsis schlegelii were overharvested to depletion, and there has been no stable supply since around 1982, despite the development of an artificial seed oyster production technique in 1975. The adverse effects of inbreeding, water pollution, and ecosystem change are some of the causes of poor growth of the artificial seed oysters (Toyama, 1991; Strack, 2006).
Lake Kasumigaura. Kazuhisa Yanase began pioneering work to cultivate 10 mm nucleated freshwater pearls at Kasumigaura in 1946. He personally tested the water, cultivated the mussels, and conducted a variety of studies, making it his life’s work to harvest pearls with wonderful color, shape, and size. In 1962, after the pearl cultivation in Lake Biwa was in decline, 10 small pearl farms moved to Lake Kasumigaura and started to culture the non-nucleated freshwater pearls (called Kasumiga keshi pearls) by using an artificial seedling Hyriopsis schlegelii mussel in a laboratory setting. The yearly production would eventually increase from a few hundred kilograms in the 1970s to more than 750 kg in the 1980s.
Also in 1962, a few pearl farmers challenged themselves to produce high-quality bead-cultured pearls over 10 mm to rival South Sea pearls and, later, to compete with mass-produced Chinese freshwater pearls, an effort which began in the late 1960s. After a thorough investigation of potential for pearl farming, their experiments proved successful in culturing spherical beaded pearls by using a new Hyriopsis hybrid, produced by crossbreeding Hyriopsis schlegelii and Hyriopsis cumingii. Initial production was only a few dozen grams per year. To minimize the risk of environmental damage to the mussels, the farms were located at estuaries of the Shintone, Sonobe, and Onogawa Rivers, which empty into Lake Kasumigaura (figure 5). The culturing of non-nucleated pearls stopped at Lake Kasumigaura at the end of the 1980s and shifted to solely nucleated culturing in 1991. From that year until 2013, average yearly production was 40–50 kg. Since 2013, production has decreased, with annual yields of less than 40 kg (R. Toda, pers. comm., 2017).

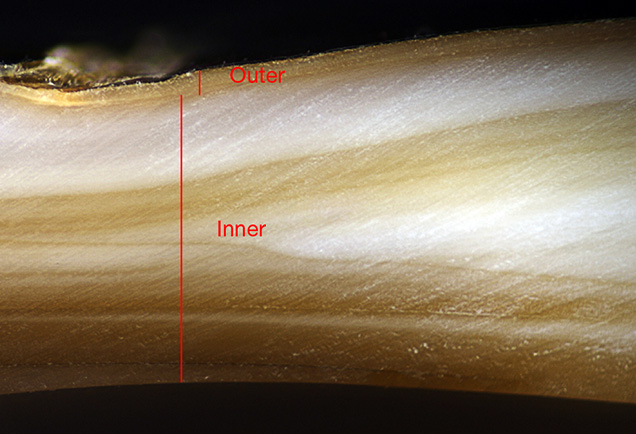
Only beaded pearls are currently produced at Lake Kasumigaura. A small mantle tissue, usually inserted into a drill hole in the bead nucleus, is inserted in each mussel, forming a sac in which a pearl starts to grow. The nacre thickness of pearls that have been cultured for longer periods (3.5–4 years) reaches 3 mm, which is much thicker than the nacre of akoya cultured pearls. The size of Kasumiga pearls typically ranges from 11 to 15 mm.
One of the characteristic features seen in Kasumiga pearls produced by the hybrid mollusk is their coloration. The variety of bodycolors includes white, pink, purple, yellow, purple-red, orange, and brown with an iridescence (Komatsu et al., 1989) that is frequently referred to as “rainbow.” Bodycolor preferences for these products differ between countries, and pink and purple colors command higher prices. In the U.S. and Europe, Kasumiga pearl is highly valued for its luster, variety of colors, and size, and the supply cannot keep up with demand.
Unfortunately, only a few pearl farms are active at Lake Kasumigaura (R. Toda, pers. comm., 2017), and the pearl farmers who operate there today do not appear to have any successors. But the Kasumiga pearl, a source of Japanese pride, will surely be able to once again thrive if the farms are able to foster successors and environmental protection measures are adopted, optimally with the government’s assistance.
PEARL CULTURING PROCESS AT LAKE KASUMIGAURA
Preparing the Donor Mantle. The farmers at Kasumigaura grow Hyriopsis cumingii × Hyriopsis schlegelii hybrid larvae in specially constructed pools for two years to protect them from any possible diseases or threats (figure 6). A scalpel is inserted into the ventral margin of the donor shell of Hyriopsis hybrid, and the anterior and posterior adductor muscles are removed to open the shell wide. Next, a long slice of mantle is cut along the pallial line using scissors. The margins are trimmed to remove pallial mantle and shape it into ribbons 3 to 4 mm wide, which are further cut into 4 × 4 mm squares (figure 7). Another method of preparing the piece, called “stripping,” was developed specifically for pearl culturing using Hyriopsis schlegelii. This method involves stripping either outer tissue that is in contact with the shell (“rear stripping”) or inner tissue that is deeper within the mussel (“front stripping” from the slice of mantle or directly from the pallial mantle). The mantle obtained by front stripping is thinner compared to rear stripping. A pearl cultured through front stripping grows faster, but the surface of the pearl shows a wrinkled appearance. Therefore, the Kasumiga pearl farms now prepare pieces mainly through rear stripping to produce pink pearls with good luster, even though they tend to take longer to grow.


Operation. Operating on freshwater cultured pearls does not require the preparation that is usually performed in saltwater pearl culturing using akoya oysters, such as high-pressure waterjet cleaning to remove any extraneous material from the surface. The shells for freshwater pearl culturing are simply washed to remove the mud from the surface the day before the operation. A nucleation operation to culture spherical pearls over 10 mm in diameter is performed by inserting a piece of mantle tissue and bead nucleus (from a U.S. freshwater mollusk of the Anodonta species, 8–8.5 mm in diameter) into a gonad that is either connected to the bottom of a digestive diverticulum or attached below a pericardial cavity. As the insertion area is not visible, the operator uses a bead that is drilled through. A metal pin is pushed through the bead with a piece of the mantle placed on the tip, to maintain a close contact between the mantle and the bead (figure 8). The operations generally take place in the winter season between October and March. They are suspended between May and September, when the water temperature exceeds 20°C or during the fertile phase, as these conditions may result in higher mortality of the mollusks. Recently, however, the mortality rate has been lowered by using antibiotics during the procedure, enabling nucleation throughout the year for some farmers.
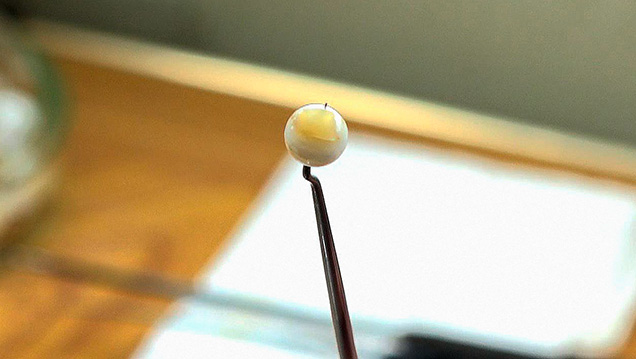
Growing Pearl Mussels. After the operation, the shells are hung for a couple of days to recover, and then they are placed in a layered net-cage and transferred to the pearl farm. The cage may have two or three layers, in which shells are neatly placed in groups of two to four with their hinges facing down (figure 9). The net-cages are hung from horizontal bamboo sticks at two-meter intervals between 70 and 100 cm below the water surface. The culturing period lasts about three to four years in general. Pearls grow between April and December, when water temperature is above 15°C. Harvesting starts gradually in the summer, in July or August, and enters the peak season in October and November, when the pearls show the most lustrous color.

SAMPLES AND ANALYTICAL METHODS
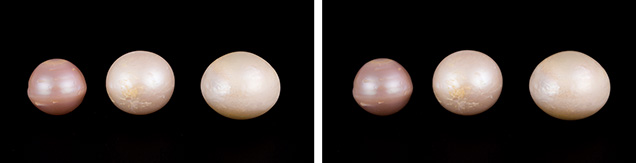
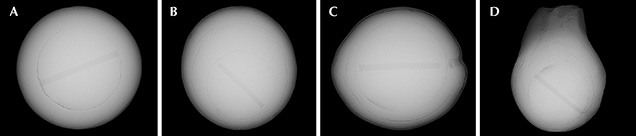
For this study, the author extracted 25 bead-cultured freshwater pearls directly from Hyriopsis hybrid shells at the Toda Shinju company’s pearl farm at Lake Kasumigaura (see table 1). The pearls ranged from round to baroque shapes with a width of 9.5–19.6 mm and a weight of 6.0–20.5 ct (figure 10). The bodycolors of the Kasumiga pearls included white, cream, light yellow, pink, purple, orange, and golden with orient. Three shells of the Hyriopsis hybrid from which the pearl samples were extracted were selected and sliced into sections for chemical composition analysis (figure 11). Chinese samples (figure 12) were also studied for comparison of chemical properties. These consisted of a Hyriopsis hybrid shell and 15 nucleated cultured pearls with 10 mm diameters, collected from the Zhong Shui Pearl Research Institute. These pearls were cultivated at Lake Taihu in Hubei Province.




The 25 pearls and three shells from Lake Kasumigaura were observed visually and microscopically for their surface growth structure and color. Their fluorescence reactions were recorded under 4-watt long-wave and short-wave ultraviolet light from a System Eickhorst MultiSpec UV-AC lamp. UV-Vis-NIR spectroscopy was performed on the surface of each sample using a Shimadzu UV-2600 spectrophotometer in diffuse reflection mode, with a resolution of 1 nm, in the 200–1000 nm range. The internal structure of the pearls was observed using a Faxitron CS-100 2D real-time X-ray microradiography (RTX) unit (90 kV and 100 mA X-ray excitation).
Raman spectra were obtained by a Horiba Jobin Yvon XploRA Plus Raman microscope equipped with a 532 nm Nd:YAG laser. The laser beam was focused through the 300 μm aperture with a 10× objective lens. The measurements were recorded on LabSpec6 software using a grating of 1800 gr/mm and spectral resolution of ±2.5 to 3.5 cm–1 between 100 and 1800 cm–1. The instrument was calibrated with a Si 520 nm reference.
Semiquantitative chemical analysis was performed using a Shimadzu EDX-8000 energy-dispersive spectrometer (EDS) equipped with an X-ray microprobe. Trace elements were measured by means of laser ablation–inductively coupled plasma–mass spectrometry (LA-ICP-MS) analysis using a Thermo Scientific iCAP RQ quadrupole ICP-MS with a ESI UP 213 Nd-YAG laser, a repetition rate of 7 Hz, an energy density of 10 J/cm2, and a spot diameter of 40 μm. The carrier gas was a mixture of helium and argon. All of the isotope ratio signals were detected, and an analytical precision (relative standard deviation 1 sigma) within 10% was achieved for all trace elements except P, which fell within 20%. Three to eight spots on each pearl sample and section of sliced shell were ablated, and the averaged data were calibrated. Microanalytical carbonate standards MACS-1 and MACS-3 pressed-powder pellets and NIST glass 610 were used as external standards.
RESULTS AND DISCUSSION
Shape and Size. Cultured pearls from Lake Kasumigaura are produced in various shapes including round, semi-round, oval, and baroque, usually with very good luster. The most common shapes are round and semi-round. Larger pearls over 16 mm have been successfully produced, but the size typically ranges between 11 and 15 mm.
Hue and Surface Structure. Cream pearls show weak luster and overtone, with irregular, interrupted, or distorted growth layers of aragonite crystals that are observed under magnification. In the studied samples, they were formed of layers of thick aragonite crystal platelets and a few black or brown organic sediments on the surface (figure 13A). Pink pearls are well produced by the Hyriopsis hybrid, showing strong luster and smooth surface reflection. They have compactly arranged layers of aragonite single crystals (figure 13B). The intensity of the pink color is determined by (1) the content of pale pigment inside the conchiolin that is a protein consisting of nacre, (2) the thickness of the aragonite single crystals, and (3) the overtone produced from the way the aragonite platelets are layered. The volume of pigment secreted differs according to the age of the shell and the water temperature, and it tends to increase when mussels are strong and healthy. Control of the shell’s physiology during culturing is the key factor to the thickness and quality of the aragonite crystal layers (Komatsu et al., 1989).
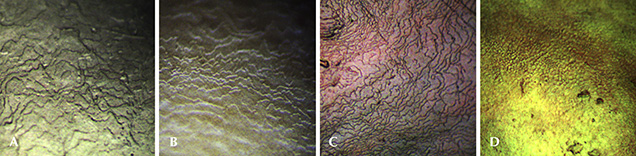
Some pink pearls are vulnerable to heat (Komatsu et al., 1989), and their color may fade slightly after one hour of exposure to strong incandescent light and heat at about 100°C (see figure 14, calibrated images before and after). Purple pearl samples showed a more violetish hue than the pink material, but with a somewhat lower luster. They contained smoothly piled growth layers of aragonite crystals with no interstitial extraneous substance (figure 13C).
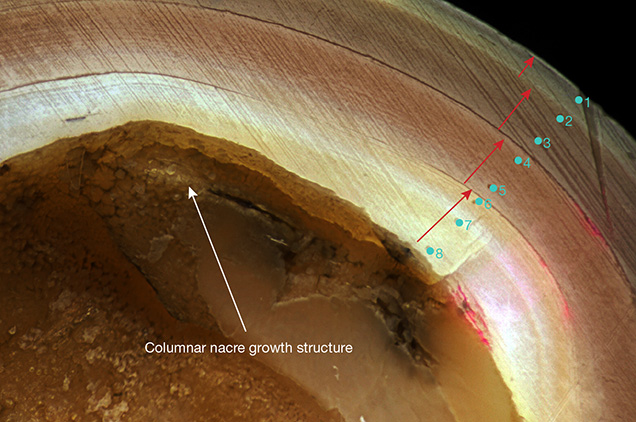
Orange samples showed a very bright yellowish orange and sometimes orange color. Observation of the cross-section revealed the sheet-laminated aragonite platelets and layers of yellow-brown pigment that had grown into the compact concentric structure (figure 15). In the inner part of the pearl, where the bead nucleus was removed to observe the initial growth structure on the nacre surface, an organic substance produced during the initial growth phase and a columnar nacre were observed. The nacre displayed four cycles of growth layers divided by white boundaries until it reached the pearl surface. This indicates a four-year culturing period, with growth interrupted in cold seasons and resuming in warmer seasons (again, see figure 15). Their color did not fade with exposure to heat.
It was learned that golden pearls with orient were actually pink-purple pearls covered with thin yellow-brown organic prismatic layers, just several dozen microns thick, across the surface (figure 13D). The golden color with orient (rainbow-like iridescence) seen on the surface is supposedly produced from an overtone mixture of laminated pink-purple aragonite crystals, an overlaid organic layer formed by abundant secretion from the shells in the growth period just before harvesting, and prismatic aragonite layers. In general, golden pearls with orient are characterized by very strong orient.
UV Fluorescence. Cream to light yellow pearl samples showed weak chalky green-blue fluorescence under long-wave UV light, whereas pink-purple pearls and golden pearls with orient showed weak chalky green fluorescence. Under short-wave UV light, the reaction became much weaker. The fluorescence appeared evenly distributed when the pearls were evenly colored. Orange pearls showed almost no fluorescence under both short-wave and long-wave UV.
UV-Vis-NIR Reflection Spectroscopy. The reflection spectra of pearls in each color are shown in figure 16. Cream pearls showed no distinct absorption except for a vague absorption band around 484 nm, while the absorption center was shifted to 498 nm in the light yellow pearls and appeared as a weak but broad absorption band. On the other hand, the purplish pink pearls colored by pigment contained in the nacre showed a large absorption band centered at around 506 nm, which became intense as the color became more saturated, with an increase in absorption between 415 and 820 nm. Study of the pearls with strong purple color revealed that general absorption between 415 and 820 nm appeared greater and the absorption band centered at 526 nm became stronger. Orange pearls showed a stronger absorption band near 498 nm, similar to light yellow pearls. The strong yellow pearls displaying rainbow iridescence that are rarely produced from the Hyriopsis hybrid showed two characteristic absorption regions. One was between the ultraviolet and the visible range at 320–460 nm with a maximum at 373 nm, and the other was in the visible range at 580–820 nm with a maximum at 640 nm.

As noted above, these reflection spectra showed an absorption band centered around 484–526 nm, indicating a feature characteristic to orange, pink, and purple pearls cultured from Hyriopsis hybrid mollusks. The absorption intensity increased with color saturation. In contrast, golden pearls with orient showed a different type of spectrum. Its golden yellow bodycolor with strong rainbow iridescent layers was supposedly produced by the absorptions in the blue and red regions between the UV and the visible range. Also, these pearls showed thick yellow-brown organic layers and prismatic layers that were formed close to the pearl surface, whereas the orange, pink, and purple pearls may be colored by pigment contained in their nacre. The author will continue to test a large number of samples to draw a detailed conclusion of the coloring pigment in pearls from Lake Kasumigaura.
Microradiography. X-ray images of an orange round (sample KSM-Ora-014), a pink near-round (KCM-Pink-013), a purple near-round (KCM-Pur-017, showing circular growth structure), and a rainbow golden baroque (KSM-GY-022) cultured pearl from Lake Kasumigaura are shown in figure 17. All the samples tested with X-ray were confirmed to contain a drilled bead, and the organic layer distributed along the boundary between the bead and nacre was very thin. The pearls shown in images A, B, and C had a similar nacre thickness, typically averaging 2 to 3 mm. Near-round pearls tended to grow a thicker nacre on one side, 1 mm on the thinner side and 5.6 mm on the thicker side. The thickest nacre observed in the baroque pearls reached approximately 8.7 mm. In the nacreous growth layer of all the pearl samples, three boundaries of organic layers were confirmed, revealing a culturing period of three years or more.
Raman Spectrum. Raman spectra of pearls with the six representative colors (cream, light yellow, pink, purple, orange, and golden with orient) are shown in figure 18. Each spectrum displays a series of peaks related to the aragonite crystal lattice at 138, 148, 187, 210, 268, and 280 cm–1; a strong peak at 1085 cm–1 that indicates symmetric stretching of (CO32–); a doublet at 701 and 705 cm–1 from plane stretching; and a weak peak at 1462 cm–1. Peaks related to the pigment (polyene, an organic compound) were confirmed at 1017, 1295, 1132, and 1522 cm–1 (Karampelas et al., 2007; Sturman et al., 2014). The peaks at 1132 (C-C single-bond stretching vibration) and 1522 cm–1 (C=C double-bond stretching vibration) tended to be weaker in cream and light yellow pearls and stronger in pink, orange, or purple pearls. The peaks at 1017 and 1295 cm–1 did not appear in the paler pearls with cream and light yellow color. The spectrum background in the golden pearls with orient was generally high, with weaker main peaks at 1085 and 701 cm–1 that indicated an aragonite crystal, confirmation that the outermost nacreous growth layers on these iridescent pearls were prismatic layers formed of aragonite, not calcite. A pigment-related peak at 1522 cm–1 was also clearly observed.

Chemical Composition. X-ray fluorescence analysis can nondestructively measure a pearl’s composition of major and trace elements to assist in the determination of some mollusk species and the culturing environment. This semiquantitative technique is also advantageous in identifying the composition of a historical or highly valuable pearl that cannot be subjected to destructive tests. In X-ray fluorescence analysis of the 25 pearl samples, CaO was detected as the main component, but organic components and water cannot be detected by this technique. A small amount of Na was detected, with an Na2O content of 1.50–5.90 wt.%. Trace elements Mn, Fe, and Sr were detected, with MnO = 0.06–0.40 wt.%, Fe2O3 = bdl–0.02 wt.%, and SrO = 0.10–0.14 wt.%. The elements Mg and K showed values close to the detection limit of the EDX-8000 spectrometer (detection limits: MgO = 0.067 wt.%, K2O = 0.016 wt.%, and Fe2O3 = 0.014 wt.%, respectively). Among these elements, Mn showed higher content in freshwater than saltwater pearls, whereas Sr tended to have higher concentration in saltwater pearls, consistent with previous studies (Wada and Fujinuki, 1976). The chemical composition results from energy-dispersive X-ray fluorescence (EDXRF) analysis indicate a slightly lower concentration compared with the results from LA-ICP-MS analysis, based on the different reference standards used in this study.
To investigate trace-element distribution using LA-ICP-MS, we ablated the surface of all 25 pearls, the section of one pearl, and the section of three Hyriopsis hybrid shells with a laser spot 40 μm in diameter, collecting analytical data on three to eight spots on each sample. The average of each detected trace element is shown in table 2.
From the LA-ICP-MS analysis on the pearls in each color and the shell from Lake Kasumigaura, minor and trace amounts of 18 elements in total (7Li, 11B, 23Na, 24Mg, 27Al, 31P, 39K, 45Sc, 47Ti, 55Mn, 57Fe, 59Co, 60Ni, 63Cu, 66Zn, 69Ga, 88Sr, and 137Ba) were consistently detected. In all the pearls tested, Na, Mn, Sr, and Fe showed rather high concentrations, ranging from 3100 to 9700 ppmw (avg. 7363 ppmw) for Na, 790–5800 ppmw (avg. 1146 ppmw) for Mn, 800–1540 ppmw (avg. 836 ppmw) for Sr, and 920–1290 ppmw (avg. 702 ppmw) for Fe. P was detected as another element of high content, between 147 and 870 ppmw. Other elements such as Mg, Al, and K showed low concentrations, between 6 and 432 ppmw. B, Ti, and Zn were below 22 ppmw, while Li, Sc, Co, Ni, Cu, and Ga were below 10 ppmw. Interestingly, Al and Ti were only detected in the outer layer (the prismatic growth area) of shell. In the analysis on the cross-section of a nacreous pearl (sample KSM-Ora-024), the nacre from the pearl’s surface to the inner side of the columnar nacre layer close to the bead nucleus showed no distinct element partition gradient except the enrichment of Na, Mg, and Fe in the columnar aragonite layer close to the bead nucleus area. Pearls produced from the Hyriopsis hybrid in Lake Kasumigaura contain less Li, Na, Mg, and Sr but much more Mn than the Japanese saltwater akoya cultured pearls from Ehime and Nagasaki Prefectures, showing the characteristic feature of freshwater species (past study data by the author). From these analysis results, there was no consistent relationship between the color of the pearls and the type and amount of transition metal elements. Thus it is reasonable to think the principal cause of color of the nacre, in the pearls or the shells, is organic pigment.
A bivalve shell is generally composed of three layers of ostracum (outer, middle, and inner). These are divided into tissue structures that include prismatic, columnar, nacreous, foliated, crossed lamellar, and complex crossed lamellar structure (Boggild, 1930; Kobayashi, 1971; Barthelat et al., 2009). The mollusk shell of the Hyriopsis hybrid from Lake Kasumigaura consists of an outer shell layer with prismatic structure and middle and inner shell layers with nacreous structure. As shown in the Raman analysis above, however, the Hyriopsis hybrid shell consists of prismatic and nacreous structures, both made of aragonite (figure 19). The shell of the Japanese akoya oyster consists of a prismatic structure made of calcite and a nacreous structure made of aragonite (Nakahara, 1994; Wada, 1999; Liu and Li, 2015). The trace-element partition of the Hyriopsis hybrid shell from Lake Kasumigaura revealed high Na and Fe contents and the trace elements Al, Ti, Co, Ni, Cu, and Zn in the outer shell layer (prismatic structure) and high Mn in the nacreous structure. To find out the relationship between the ostracum of a shell and the trace-element contents, the factors in the secretion function in a tissue or in ostracum that control the content of trace elements should be analyzed in the future. The content of trace elements such as B, Mg, and P seemed to be somewhat lower in the shell than in the pearls.
Large amounts of Chinese freshwater bead-cultured pearls showing Kasumiga-like colors have recently been supplied to the global market. We analyzed 15 beaded freshwater pearls in each color (cream, light yellow, pink, purple, and orange) and the cross-section of a Hyriopsis hybrid shell from Hubei to compare the features of trace elements.
LA-ICP-MS analysis detected distinctly higher amounts of Ba and Ga in the Chinese pearls of all six colors and in the shell. The content ranges of other metallic elements such as B, Mg, K, Mn, Fe, Sr, Co, Ni, Cu, and Zn were similar to those of Kasumiga pearls. The analyzed chemical data were plotted on the chemical fingerprint diagram in figure 20 using a combination of the trace elements Ba and Ga to differentiate the freshwater pearls and shells of the Hyriopsis hybrid from both Japan and China. Ba and Ga values for the freshwater pearls and the Hyriopsis hybrid shell from Kasumigaura were concentrated in the lower region and clearly distinguished from the Chinese counterparts from Lake Taihu without overlap. Despite the use of the same species of Hyriopsis hybrid oysters in both countries, they showed different concentrations of heavy metal ions (Ba2+, Ga2+) in both the pearls and the shells, presumably in direct proportion to that of the aquatic habitat, as those ions are contained in extracellular fluid in the mantle. The application of this diagram to freshwater pearls and shells from China or other countries is subject to validation in future studies.

Future Outlook for the Kasumiga Pearl Industry. Since the late 1980s, the mass production and inexpensive price of Chinese freshwater cultured pearls has transformed the Japanese pearl industry. Although it is almost impossible to compete with China in the present circumstances, a few farmers are dedicating their efforts to improving culturing techniques, growing shells with great care, and strictly monitoring water quality, prioritizing quality over mass production. Kasumiga pearls started attracting attention from Europe and the United States in the 1990s but are scarcely sold in Japan’s domestic market. Improvements in the quality of the pearls, as well as stronger marketing and branding, are critical to eliminating cheap Kasumiga products and maintaining a higher price point.
CONCLUSIONS
The production of nucleated freshwater cultured pearls from Lake Kasumigaura has subsided in recent years, but large Kasumiga pearls with thick nacre and strong luster in pink or purple colors are highly valued in the international market. In this study, the author visited the Toda Shinju pearl farm at the Ono River, which empties into Lake Kasumigaura, to harvest pearls and shells directly and analyze them. The pearls were analyzed by means of absorption and Raman spectroscopy, observation of internal structure, and quantitative compositional analysis for major and trace elements.
The freshwater nucleated cultured pearls collected from Hyriopsis hybrid shells were divided into six groups of representative colors: cream, light yellow, pink, purple, orange, and golden with orient. The nacreous growth layer in the cream and light yellow pearls showed thick layers of aragonite platelets and laminated irregularity with low tone, and a vague absorption band between 480 and 500 nm in the visible region of the UV-Vis-NIR reflectance spectrum. Pink, purple, and orange pearls demonstrated the nacre’s very fine crystalline structure with neat lamination and very good luster. They showed a strong absorption center in the range between 480 and 530 nm, which increased in intensity as the color tone and saturation of the pearl became stronger, marking the characteristic increment in a wider range between 415 and 820 nm. The surface of the golden pearls with orient was covered with a thin prismatic layer of aragonite that was rich in yellow-brown organic substance, and these pearls were characterized by strong orient. They showed two absorption regions in the visible range, one at 320–460 nm centered at 373 nm and the other at 580–820 nm centered at 640 nm. Raman spectroscopy detected a pigment contained in the nacre.
Kasumiga pearls generally do not undergo treatments such as bleaching and dyeing, and the samples showed very weak chalky green-blue fluorescence or no reaction under UV irradiation, providing proof of natural color.
Semiquantitative analysis of the Kasumiga pearls detected a main component of CaO, followed in descending order by Na, Mn, Sr, and Fe. LA-ICP-MS analysis detected 18 minor and trace elements, of which Na, Mn, Sr, and Fe were in the highest concentration (over 1000 ppmw). P, Mg, Al, and K were in the second-highest group (several hundred to several dozen ppmw). B, Ti, and Zn were under 22 ppmw, while Li, Sc, Co, Ni, Cu, and Ga were detected in trace amounts (under 10 ppmw). Al and Ti were only distributed in the prismatic aragonite growth layer on the outer part of the mollusk shell. All the metallic elements detected were confirmed to be unrelated to the color of the pearls.
In Lake Taihu, China, large volumes of freshwater beaded cultured pearls using Hyriopsis hybrid shells of the same species used at Lake Kasumigaura have been produced for the international market. Chemical analysis by LA-ICP-MS demonstrated that chemical fingerprinting using the Ba to Ga ratio was effective in distinguishing freshwater nucleated pearls from these two countries.



