

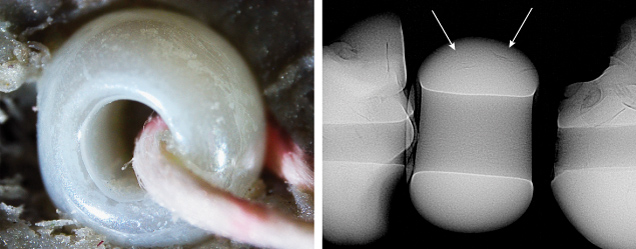
GIA’s Mumbai laboratory recently received a submission of 18 strands of pearls for identification, consisting of approximately 9,360 white to light cream pearls weighing a total of 706.79 carats. When subjected to X-ray fluorescence, most of the pearls were inert, indicating a saltwater origin, while a few exhibited the strong yellowish green fluorescence of freshwater pearls. Notably, one white oval “pearl,” measuring approximately 1.70 × 1.12 mm and weighing 0.01 ct (figure 1), displayed a unique moderate blue reaction upon X-ray exposure (figure 2)—an effect not previously observed in saltwater or freshwater pearls. The “pearl” was visually similar to the other submitted pearls, but its extremely large drill hole raised suspicion (figure 3, left). Under higher magnification, its smooth surface lacked characteristic nacreous overlapping aragonite platelets, displaying a unique liquid-like surface residue instead. This “pearl” appeared inert under long-wave ultraviolet light, while other pearls were greenish yellow. Additionally, it exhibited a light yellow reaction under the deep-UV wavelength (<225 nm) of the DiamondView, unlike the typical blue reactions observed in pearls. Raman spectroscopy with different laser excitations did not reveal any distinctive features indicative of calcium carbonate polymorphs typically found in pearls. Based on these findings, it was concluded that the material in question was not a pearl but rather an imitation. Real-time X-ray microradiography (RTX) indicated that, with the exception of the one imitation, the 18 strands contained a mixture of natural and non-bead cultured pearls. Interestingly, the imitation displayed a similar radiopacity to the other pearls. A few arcs were present at the outer edges, comparable to growth arcs observed in the adjacent natural pearls strung in the strand (figure 3, right).
Common pearl imitations often utilize material such as solid or hollow glass, plastic, or shell bead coated with a “pearl essence” substance that simulates nacre’s iridescence (J. Hanano et al., “Majorica imitation pearls,” Fall 1990 G&G, pp. 178–188). These imitations lack the usual nacreous platelets, mosaic patterns, and cellular structures observed in pearls, and often present a glittery surface appearance of coating material and occasional “molded” drill holes. Such imitations may show variations in RTX results depending on the composition of the substance and the coating.
Distinguishing imitations when mixed with large quantities of pearls in an item can be a challenge. Due to their similarity in appearance, these materials are occasionally mixed with pearl strands and jewelry. Furthermore, the presence of arcs in the examined imitation sample may have been misleading, potentially going unnoticed without the aid of the blue fluorescence under X-ray exposure during routine testing. The utilization of X-ray fluorescence screening has proven useful in the identification of certain types of imitation materials, particularly in the case of smaller pearls strung in strands. While encountering such imitation materials is not frequent in routine testing, it remains essential to accurately distinguish and identify them to maintain the authenticity of pearl collections.


GIA’s Mumbai laboratory recently received a submission of 18 strands of pearls for identification, consisting of approximately 9,360 white to light cream pearls weighing a total of 706.79 carats. When subjected to X-ray fluorescence, most of the pearls were inert, indicating a saltwater origin, while a few exhibited the strong yellowish green fluorescence of freshwater pearls. Notably, one white oval “pearl,” measuring approximately 1.70 × 1.12 mm and weighing 0.01 ct (figure 1), displayed a unique moderate blue reaction upon X-ray exposure (figure 2)—an effect not previously observed in saltwater or freshwater pearls. The “pearl” was visually similar to the other submitted pearls, but its extremely large drill hole raised suspicion (figure 3, left). Under higher magnification, its smooth surface lacked characteristic nacreous overlapping aragonite platelets, displaying a unique liquid-like surface residue instead. This “pearl” appeared inert under long-wave ultraviolet light, while other pearls were greenish yellow. Additionally, it exhibited a light yellow reaction under the deep-UV wavelength (<225 nm) of the DiamondView, unlike the typical blue reactions observed in pearls. Raman spectroscopy with different laser excitations did not reveal any distinctive features indicative of calcium carbonate polymorphs typically found in pearls. Based on these findings, it was concluded that the material in question was not a pearl but rather an imitation. Real-time X-ray microradiography (RTX) indicated that, with the exception of the one imitation, the 18 strands contained a mixture of natural and non-bead cultured pearls. Interestingly, the imitation displayed a similar radiopacity to the other pearls. A few arcs were present at the outer edges, comparable to growth arcs observed in the adjacent natural pearls strung in the strand (figure 3, right).

Common pearl imitations often utilize material such as solid or hollow glass, plastic, or shell bead coated with a “pearl essence” substance that simulates nacre’s iridescence (J. Hanano et al., “Majorica imitation pearls,” Fall 1990 G&G, pp. 178–188). These imitations lack the usual nacreous platelets, mosaic patterns, and cellular structures observed in pearls, and often present a glittery surface appearance of coating material and occasional “molded” drill holes. Such imitations may show variations in RTX results depending on the composition of the substance and the coating.
Distinguishing imitations when mixed with large quantities of pearls in an item can be a challenge. Due to their similarity in appearance, these materials are occasionally mixed with pearl strands and jewelry. Furthermore, the presence of arcs in the examined imitation sample may have been misleading, potentially going unnoticed without the aid of the blue fluorescence under X-ray exposure during routine testing. The utilization of X-ray fluorescence screening has proven useful in the identification of certain types of imitation materials, particularly in the case of smaller pearls strung in strands. While encountering such imitation materials is not frequent in routine testing, it remains essential to accurately distinguish and identify them to maintain the authenticity of pearl collections.




